the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
A case study of developmental palaeontology in Stereosternum tumidum (Mesosauridae, Parareptilia)
Constanze Bickelmann
Linda A. Tsuji
Ontogenetic series of extinct taxa are rare. However, if preserved, fossil embryos and juveniles can provide evidence of developmental plasticity as related to ecological specialization. Here, we describe articulated and isolated juvenile material found in close association with an adult mesosaurid Stereosternum tumidum (MB.R.2089) from Lower Permian sediments in Brazil, housed in the collection of the Museum für Naturkunde Berlin. Stylopodial, zeugopodial, and autopodial elements are not yet completely ossified in the juveniles, as indicated by compression artifacts on the surface of the bone. These correspond to internal ossification processes, which have been demonstrated in other aquatic taxa. Quantitative analysis of measurements in juvenile and adult material reveals differing growth rates between limb elements: hind limb zeugopodia, which are massive and elongate in the adult as needed for propulsion, are already comparatively larger in the juvenile than the humeri, femora, and also the zeugopodia of the forelimb. This pattern differs from that seen in another extinct aquatic reptile, Hovasaurus boulei. Nevertheless, we attribute the accelerated growth rate or earlier onset of ossification to be a potential developmental pathway generating limb element variation in the adult present in 280 million year old mesosaurs, which are known for their fully aquatic lifestyle, in which the hind limbs play a more prominent role than the forelimbs.
- Article
(20453 KB) - Full-text XML
- BibTeX
- EndNote
The study of fossilized ontogenies is largely confined to postnatal stages with ossified skeletal elements, due to preservational (taphonomic) factors. If preserved, they provide valuable insight into the life history of extinct taxa, as well as evolutionary trajectories. To date, ossification sequences in fossils are available for some “fish” (Cloutier, 2010), temnospondyl and lepospondyl amphibians (Fröbisch, 2008; Fröbisch et al., 2010, 2015), “younginiforms” (sensu Bickelmann et al., 2009) (Currie, 1981; Caldwell, 2002), mosasaurs (Caldwell, 2002), sauropterygians (Hugi and Scheyer, 2012), ichthyosaurs (Caldwell, 1997), sauropsids (Delfino and Sánchez-Villagra, 2010) and mammals (Sánchez-Villagra, 2010). Most of this information is derived from secondarily aquatic taxa, which is the result of a taphonomic bias (Fröbisch et al., 2010). A notable lack of published developmental data for early amniote taxa makes each incidence a valuable contribution to our knowledge of the patterns. Gaining a comprehensive picture of changes in development and growth in extinct (fossil) taxa in addition to what we know about development in modern animals, as approached in this and similar studies, contributes to our understanding of evolution, developmental trajectories, life history strategies, and more (Fröbisch et al., 2010).

Figure 1Overview photographs of rock slab (MB.R.2089.A, a) and latex cast (MB.R.2089.B, b) of Stereosternum tumidum. For detail (boxes) see Fig. 2. Scale bar equals 50 mm. For abbreviations see Material and Methods section.
Mesosauridae (Reptilia: Parareptilia) is an extinct secondarily aquatic group from the Paleozoic; notably, their fossil record also includes ontogenetic data (Rieppel, 1993; Piñeiro et al., 2012a). The clade consists of the three monospecific genera Mesosaurus tenuidens, Stereosternum tumidum, and Brazilosaurus sanpauloensis from Lower Permian localities in Brazil and southern Africa (Oelofsen and Araújo, 1983, 1987). Their elongate skulls, slender teeth, paddle-like limbs, and thickened (pachyostotic) trunk ribs are indicative of a secondarily fully aquatic lifestyle and distinguish mesosaurs from all other Paleozoic amniotes (Modesto, 2006). Recent studies suggest that mesosaurs, or at least Mesosaurus, were slow swimmers living in shallow lagoon-like waters, possibly filter-feeding on pygocephalomorph crustaceans (Piñeiro et al., 2012b; Villamil et al., 2016). Hypothesized viviparity in Mesosaurus is another potential indicator of a fully aquatic lifestyle (Piñeiro et al., 2012a). Most recent studies suggest mesosaurs group basally within parareptiles; a small amniote clade that originated in the Late Carboniferous (Tsuji and Müller, 2009; Modesto et al., 2015; though see contradictory hypothesis of Laurin and Piñeiro, 2017). Evolutionary novelties in this clade include impedence-matching hearing in nycteroleterids (Müller and Tsuji, 2007), caudal regeneration in mesosaurs (Delfino and Sánchez-Villagra, 2010), derived feeding mechanisms and bipedality in bolosaurids (Reisz et al., 2007; Berman et al., 2000), and secondarily aquatic lifestyles as well as (ovo)viviparity in mesosaurids (Modesto, 2006; Piñeiro et al., 2012a), all of which occurred in parareptiles as early as the Early Permian. The first occurrence of congenital scoliosis is also reported from members of this clade (Szczygielski et al., 2017). Fortunately, there is some ontogenetic data available for parareptiles, such as in the pareiasaurs Elginia (Spencer and Lee, 2000) and Deltavjatia (Tsuji, 2013), procolophonids (Cisneros, 2008), and Mesosaurus (Rieppel, 1993; Piñeiro et al., 2012a).
Here, we describe a remarkable specimen containing an adult Stereosternum tumidum next to juvenile material from Lower Permian sediments in Brazil; a specimen long neglected in the fossil collections at the Museum für Naturkunde Berlin. Parent–hatchling associations in mesosaurs have thus far only been described for the sister-taxon Mesosaurus (Piñeiro et al., 2012a). The present finding of differing developmental stages allows for the establishment of baseline ossification sequences and growth rates. This study proposes that they relate to the adaptation of mesosaur limbs to a fully aquatic lifestyle.
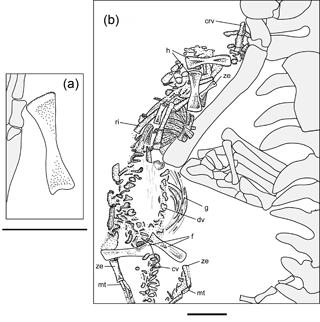
Specimen MB.R.2089 consists of a natural mold (MB.R.2089.A), preserving an articulated adult postcranium and the articulated postcranial elements of a juvenile, as well as an isolated long bone from another juvenile (Fig. 1). A latex cast (MB.R.2089.B) was prepared at the Museum für Naturkunde Berlin in 2013. In this, the adult is visible in ventral aspect. It was not until the latex cast was made that the juvenile skeleton was recognized as such, and the majority of the anatomical observations of this smaller skeleton were made directly from the cast. A historical label applied to the specimen assigned the adult to the taxon Stereosternum tumidum, an identification we verify using the anatomical features listed below. The close proximity of the juvenile material to the adult on the rock slab strongly suggests kinship (Piñeiro et al., 2012a).
Systematic Paleontology
Parareptilia Olson, 1947
Mesosauridae Baur, 1889
Stereosternum tumidum Cope, 1885
Type specimen: MB.R.2089
Locality and horizon: Itapetininga, Sao Paolo, Brazil. Passa Dois Group.
Irati Formation. Lower Permian. Artinskian. Permian 2.
3.1 Description of the adult
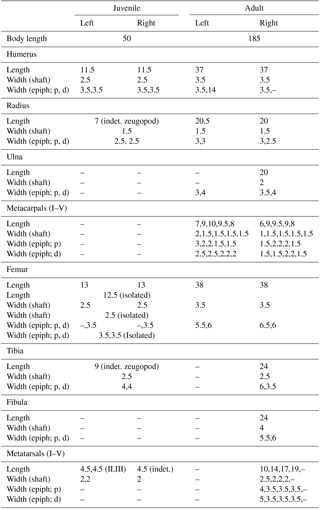
The adult consists of a well-preserved and articulated postcranium including presacral, sacral, and approximately 23 caudal vertebrae, cervical and abdominal ribs (generally 5mm thick in the midshaft region), as well as both complete fore- and hind limbs with pectoral and pelvic girdle (Fig. 1). The skull is absent (Fig. 1). All observable aspects of the postcranial anatomy conform to that of Stereosternum tumidum, as described by Modesto (1999, 2010). Specifically, the unfused intermedium and lateral centrale, the presence of a pisiform, and a diamond-shaped interclavicle characterize this specimen as Stereosternum rather than Mesosaurus (Fig. 1; Modesto, 1999, 2010). The fore- and hind limbs are elongate, characteristic for the aquatic lifestyle proposed for mesosaurs (Modesto, 2006; Piñeiro et al., 2012a, b; Villamil et al., 2016). Size and appearances of the fore- and hind limb stylopodia are similar, although the femora are slightly shorter. The ratio of the midshaft diameter to total length of the femur is around 9.2 %, which lies between an earlier erected 11 % in Stereosternum as compared to around 8 % for Mesosaurus (Modesto, 1999, 2010). Zeugopodial and autopodial elements of the hind limbs are wider in the midshaft region and the proximal epiphyses compared to the ones in the forelimbs (Table 1). All limb measurements are listed in Table 1.
3.2 Description of the juvenile material
The juvenile material preserves one mostly complete individual and an isolated long bone element. Although there is considerable intraspecific variation in limb element length and shape in sub-adult mesosaurs (Rossmann and Maisch, 1999), we believe the isolated element belonging to a second individual of Stereosternum is most likely a femur based on size and appearance of the element compared to that of the mainly complete individual (Fig. 2a; Table 1).
The almost complete individual consists of elements of the postcranium, including vertebral column, ribs, gastralia, and fore- and hind limbs (Fig. 2b). The skull is not visible; it may be present in the rock in an underlying layer, or may have been separated from the postcranial on the missing counterpart (Fig. 2b). Body size, if measured from the approximate position of last cervical to first sacral rib, is approximately 30 % of the size of the adult (Table 1). The taphonomic orientation of the skeleton cannot be attributed, as the elements are typically in close association but not completely articulated, but the axis of the skeleton is oriented in the same direction as in the adult and is “stretched out”, rather than curled into a foetal position (Fig. 2b). The mostly complete juvenile is located close to the right hind limb of the adult skeleton and does not appear to have been greatly disturbed pre or post burial, in contrast to the isolated femur, which lies close to the adult left forelimb. Although the skeletal elements are not completely articulated as can be seen in the adult, they are very closely associated and in their approximate anatomical positions.
Cervical vertebrae, located proximal to the forelimbs, are clearly diagnosable. However, dorsal and caudal vertebrae are distinguishable due only to their location in situ (Fig. 2b). While cervical vertebrae are fully developed, the neural arch and pleurocentra of both dorsal and caudal vertebrae are not yet fully fused and are preserved in two adjacent lines, though the lack of definitive anatomy in these bones makes it difficult to identify any individual element (Fig. 2b). Concerning length, the small skeleton retains what appears to be an almost complete tail, and at least 25 vertebrae can be recognized (Fig. 2b). Further caudally, vertebral elements are more difficult to discern, yet there appears to be the continuation of the outline of the soft parts of the tail with occasional bone fragments visible. Adult Mesosaurus and Brazilosaurus typically have more than 60 caudal vertebrae (Modesto, 2010), so it is reasonable to conjecture that the caudal vertebrae in this juvenile Stereosternum numbered more than what can be directly observed here. The ribs are already diagnostically thickened (pachyostotic; most are 2 mm thick in the midshaft) in the juvenile and are scattered throughout the thoracic region (Fig. 2b). Slender gastralia lie in the abdominal region (Fig. 2b).
Identifiable juvenile limb elements preserved in MB.R.2089 include two humeri, three femora, one indeterminate forelimb zeugopodial element, one indeterminate hind limb zeugopodial element, and three metatarsals. Due to the extreme delicacy of the juvenile skeleton, other limb elements may be preserved but obscured under those we can identify, or not differentiated enough from the “matrix” to definitively name. In the forelimb, both humeri do not exhibit characteristics such as the deltopectoral crest, ectepicondylar groove, entepicondylar foramen, and an enlarged entepicodyle that are present in the adult (Figs. 1, 2b). Instead, they retain metaphyseal compression and epiphyseal ridges (Fig. 2b) (sensu Hugi and Scheyer, 2012), indicating low maturity similar to what is seen in pachypleurosaur juveniles (Hugi and Scheyer, 2012). In the hind limbs, all femora show reduced ontogenetic compression (sensu Hugi and Scheyer, 2012) compared with the humeri, yet also lack adult phenotypic features such as patellar groove and muscle attachment sites for trochanter (Fig. 2a, b). One of either the fibula or tibia can be identified per side (Fig. 2b, Table 1). Due to the close association of the zeugopod and the preserved metatarsals, we hypothesize that tarsals are not yet ossified (Fig. 2b), a condition also seen in other juvenile mesosaur skeletons (i.e. BSP 1979, fig. 4 from Rossman and Maisch, 1999). Two faint metatarsals can be identified in the latex cast (MB.R.2089.B). According to their location, we think these might be the middle ones (II & III; Fig. 2b, Table 1). On the other side, one indeterminate metatarsal is preserved (Fig. 2b, Table 1).
3.3 Growth Rates
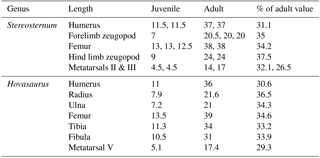
We calculated growth rates using limb measurements in Table 1. We find that the juvenile hind limb zeugopod, which is longer in the hind limb as compared to the forelimb in the adult, is already slightly larger (37.5 % of the adult size) than all other limb elements (ranging from 26.5 % in the metatarsal III to 35 % in the forelimb zeugopod), indicating a faster growth rate or an earlier onset of ossification (Table 2).
Further, we compared the established growth rates in MB.R.2089 to earlier published limb growth data from another secondarily aquatic reptile, Hovasaurus boulei from the Upper Permian of Madagascar (Currie, 1981). Hovasaurus is a “younginiform” diapsid reptile (sensu Bickelmann et al., 2009), for which, fortunately much ontogenetic material is available (Currie, 1981). It also displays enlarged hind limbs compared to the forelimbs, which are potentially useful when swimming (Currie, 1981). Comparing the ossification of limb elements in sub-adults and adults of Stereosternum and Hovasaurus, we find that the humerus, forelimb zeugopod, femur, and metatarsals show growth rates or onsets of ossification to be comparable (Table 2). However, the growth rate of the hind limb zeugopod is higher in Stereosternum compared to Hovasaurus (Table 2).
The secondarily fully aquatic parareptilian clade Mesosauridae is renowned for an exceptional fossil record including the preservation of multiple growth stages, tail regeneration, along with the earliest report of (ovo)viviparity in amniotes (Rieppel, 1993; Delfino and Sánchez-Villagra, 2010; Piñeiro et al., 2012a). Here, we describe a congregation of adult and sub-adult (or potentially hatchling) material of the taxon Stereosternum tumidum. Parent-hatchling associations have been previously reported in mesosaurs only in Mesosaurus tenuidens (Piñeiro et al., 2012a).
Ontogenetic series of fossils provide valuable insights into life history traits of organisms (Fröbisch et al., 2010). Based on the described juvenile material of Stereosternum we can make two observations:
-
Ossification sequences. Generally in tetrapods, stylopod ossification starts before that of the zeugopod and autopod, and mesopodial (wrist and ankle) ossification is significantly delayed in relation to all other limb elements (Rieppel, 1993). In the articulated juvenile mesosaur, the mesopodial elements are not yet ossified (Fig. 2b). In another isolated juvenile, attributed to Stereosternum (NHMUK R3521), the astragalus is ossified but not the calcaneum. Therefore, Stereosternum apparently follows the same ossification pattern in their mesopodia as the sister-taxon Mesosaurus (Rieppel, 1993).
-
Growth rates. Mesosaurs display aquatic specializations in their limbs, with enlarged zeugo- and autopodia in the hind limbs compared to those of the forelimbs (Rossmann and Maisch, 1999). This size variation is also present in the adult in MB.R.2089 (Fig. 1, Table 1).
First, taking the juvenile material into account, we calculated growth rates for various limb elements in Stereosternum tumidum (Table 2). Results show that the hind limb zeugopodia are more highly ossified, as demonstrated by the greater longitudinal length of the elements (37.5 %) of the sub-adult as compared to the humeri, femora, forelimb zeugopodia, and metatarsals (ranging from 26.5 to 35 %; Table 2). We associate this accelerated growth rate or earlier onset of ossification to the more robust morphology of the adult tibiae and fibulae (see epiphyseal widths in Table 1). This pattern is potentially related to stronger hind limb bones needed for propulsion during swimming. Such a phenomenon has been demonstrated for secondarily aquatic skinks (Hugi et al., 2012). Microanatomical analyses would help shed light on its internal strength parameters, such as bone density and cortical thickness (Houssaye et al., 2016), but are not possible with the present natural mold. Unfortunately, because no metacarpals are preserved, no predictions can be made about such a pattern in the autopodia.
Secondly, in order to test for potential heterochronies, we established limb growth ratios for another fossil reptile, Hovasaurus boulei from the Upper Permian of Madagascar (Currie, 1981). Limb lengths and proportions in adult Hovasaurus are in most respects very similar to those of Stereosternum (MB.R.2089). However; in Hovasaurus, the hind limb zeugopod is not only substantially longer than that of the forelimb in this taxon, it is also longer than the hindlimb zeugopod of Stereosternum (Currie, 1981). In fact, growth rates of the humeri, femora, forelimb zeugopodia, and metatarsals in Stereosternum and Hovasaurus are more or less similar (Table 2). However, growth rates for the hind limb zeugopodia show a significant difference: in Stereosternum the juvenile elements have already reached 37.5 % of the adult length, in contrast to only 33.2–33.9 % in Hovasaurus (Table 2). This accelerated growth rate or earlier onset of ossification in Stereosternum is surprising given that hind limb zeugopodia in adult Hovasaurus are even longer than in adult Stereosternum. Still, we suggest that this finding of skeletal heterochrony is related to the aquatic lifestyle in Mesosauridae. In other extinct marine reptiles, hyperdactyly and hyperphalangy were other adaptations to a fully aquatic lifestyle (Caldwell, 2002). The relevant pattern in Hovasaurus remains to be characterized.
Heterochronies (evolutionary shifts in developmental timing) are an important tool for morphological change in the phenotype; e.g. limb allometries result from differences in the growth patterns of skeletal elements (Richardson, 1999). In Stereosternum, the advanced growth rate or earlier onset of ossification in hind limb zeugopodia as compared to stylopodia and forelimb zeugopodia, can be linked with longer zeugopodia and autopodia in the adult and are secondarily related to a fully aquatic lifestyle (Rossmann and Maisch, 1999). The autopodia in MB.R.2089 are not well preserved and thus can not be evaluated here. This phenomenon, in which selection for an adult trait produces crucial changes early in ontogeny, is called developmental penetrance (Richardson, 1999). In fact, differences in the phenotype are generated at a variety of ontogenetic stages (Richardson, 1999). Other mechanisms include changes at the cartilaginous level, e.g. in fossorial talpid moles in which the cartilage anlage is already a miniature of the adult phenotype including its distinct ecomorphological specializations, as related to their extreme digging behaviour (Bickelmann et al., 2014). Transcriptional heterochrony (changes in the spatial and temporal expression of developmental genes during ontogeny) is another developmental mechanism leading to morphological variation at the molecular level (Richardson et al., 2009; Bickelmann et al., 2012). Here, only subtle changes are needed to produce a significantly different phenotype (Richardson, 1999). However, these are difficult, if not impossible, to trace in the fossil record.
Our study shows evidence that differential growth rates and skeletal heterochrony are developmental trajectories that could potentially account for phenotypic variation in adult limb morphology, in this case related to adaptation to a fully aquatic lifestyle in an extinct tetrapod clade as early as 290 mya.
No data sets were used in this article.
| as | astragalus |
| ca | calcaneum |
| crv | cervical vertebra |
| cv | caudal vertebra |
| dc | distal carpals |
| dpc | deltopectoral crest |
| dt | distal tarsals |
| dv | dorsal vertebra |
| ect | ectepicondyle |
| ent | entepicondyle |
| ent f | entepicondylar foramen |
| f | femur |
| fi | fibula |
| g | gastralia |
| h | humerus |
| i | intermedium |
| icl | interclavicle |
| il | ilium |
| is | ischium |
| lc | lateral centrale |
| mc | metacarpal |
| mt | metatarsal |
| p | phalanx |
| pg | patellar groove |
| pi | pisiform |
| pu | pubis |
| r | radius |
| ri | rib |
| s | sacral rib |
| sc | scapulacoracoid |
| t | tibia |
| u | ulna |
| ul | ulnare |
| ze | zeugopodial element |
| MB | Museum für Naturkunde Berlin, Germany |
| MNHN | Muséum National d'Histoire Naturelle, Paris, France |
| NHMUK | Natural History Museum, London, UK |
| SAM | South African Museum, Cape Town, South Africa |
CB and LAT contributed equally to the study design, data analysis, and writing of the manuscript.
The authors declare that they have no conflict of interest.
We thank Henrik Stöhr (Tübingen) for the
preparation of the latex cast. Carola Radke (Berlin) is thanked for taking
the photographs. Thanks to Oliver Hampe (Berlin) for access to the
collection under his care. We acknowledge constructive criticism by two
anonymous reviewers and the editor, which improved an earlier version of the
manuscript. This work was funded by the German Research Foundation (DFG), BI 1750/3-1 for Constanze Bickelmann.
Edited by: Johannes Müller
Reviewed by: two anonymous referees
Baur, G.: On the morphology of the vertebrate-skull, J. Morphol., 3, 467–474, 1889.
Berman, D. S., Reisz, R. R., Scott, D., Henrici, A. C., Sumida, S. S., and Martens, T.: Early Permian Bipedal Reptile, Science, 290, 969–972, 2000.
Bickelmann, C., Müller, J., and Reisz, R. R.: The enigmatic diapsid Acerosodontosaurus piveteaui (Reptilia: Neodiapsida) from the Upper Permian of Madagascar and the paraphyly of “younginiform” reptiles, Can. J. Earth Sci., 46, 651–661, 2009.
Bickelmann, C., Mitgutsch, C., Richardson, M. K., de Bakker, M. A. G., Jiménez, R., and Sánchez-Villagra, M. R.: Transcriptional heterochrony in talpid mole autopods, EvoDevo, 3, 16, 2012.
Bickelmann, C., Jiménez, R., Richardson, M. K., and Sánchez-Villagra, M. R.: Humerus development in moles (Talpidae, Mammalia), Acta Zool.-Stockholm, 95, 283–289, 2014.
Caldwell, M. W.: Limb ossification patterns of the ichthyosaur Stenopterygius, and a discussion of the proximal tarsal row of ichthyosaurs and other neodiapsid reptiles, Zool. J. Linn. Soc.-Lond., 120, 1–25, 1997.
Caldwell, M. W.: From fins to limbs to fins: limb evolution in fossil marine reptiles, Am. J. Med. Gen., 112, 236–249, 2002.
Cisneros, J. C.: Phylogenetic relationships of procolophonid parareptiles with remarks on their geological record, J. Syst. Palaeontol., 6, 345–366, 2008.
Cloutier, R.: The fossil record of fish ontogenies: Insights into developmental patterns and processes, Semin. Cell Dev. Biol., 21, 400–413, 2010.
Cope, E. D.: Twelfth contribution to the herpetology of tropical America, P. Am. Philos. Soc., 22, 167–194, 1885.
Currie, P. J.: Hovasaurus boulei, an aquatic eosuchian from the Upper Permian of Madagascar, Palaeontologia Africana, 24, 99–168, 1981.
Delfino, M. and Sánchez-Villagra, M. R.: A survey of the rock record of reptilian ontogeny, Semin. Cell Dev. Biol., 21, 432–440, 2010.
Fröbisch, N. B.: Ossification patterns in the tetrapod limb – conservation and divergence from morphogenetic events, Biol. Rev., 83, 571–600, 2008.
Fröbisch, N. B., Olori, J. C., Schoch, R. R., and Witzmann, F.: Amphibian development in the fossil record, Semin. Cell Dev. Biol., 21, 424–431, 2010.
Fröbisch, N. B., Bickelmann, C., Olori, J. C., and Witzmann, F.: Deep-time evolution of regeneration and preaxial polarity in tetrapod limb development, Nature, 527, 231–234, 2015.
Houssaye, A., Sander, P. M., and Klein, N.: Adaptive patterns in aquatic amniote bone microanatomy – more complex than previously thought, Integr. Comp. Biol., 56, 1349–1369, 2016.
Hugi, J. and Scheyer, T. M.: Ossification sequences and associated ontogenetic changes in the bone histology of pachypleurosaurids from Monte San Giorgio (Switzerland/Italy), J. Vertebr. Paleontol., 32, 315–327, 2012.
Hugi, J., Hutchinson, M. N., Koyabu, D., and Sánchez-Villagra, M. R.: Heterochronic shifts in the ossification sequences of surface-and subsurface-dwelling skinks are correlated with the degree of limb reduction, Zoology, 115, 188–198, 2012.
Laurin, M. and Piñeiro, G. M.: A reassessment of the taxonomic position of Mesosaurs, and a surprising phylogeny of early amniotes, Front. Earth Sci., 5, 88, https://doi.org/10.3389/feart.2017.00088, 2017.
Modesto, S. P.: Observations on the structure of the Early Permian reptile Stereosternum tumidum Cope, Palaeontologia Africana, 35, 7–19, 1999.
Modesto, S. P.: The cranial skeleton of the Early Permian aquatic reptile Mesosaurus tenuidens: implications for relationships and palaeobiology, Zool. J. Linn. Soc., 146, 345–368, 2006.
Modesto, S. P.: The postcranial skeleton of the aquatic parareptile Mesosaurus tenuidens from the Gondwanan Permian, J. Vertebr. Paleontol., 30, 1378–1395, 2010.
Modesto, S. P., Scott, D. M., MacDougall, M. J., Sues, H.-D., Evans, D. C., and Reisz, R. R.: The oldest parareptile and the early diversification of reptiles, Proceedings B, 282, 20141912, https://doi.org/10.1098/rspb.2014.1912, 2015.
Müller, J. and Tsuji, L. A.: Impedance-matching hearing in Paleozoic reptiles: evidence of advanced sensory perception at an early stage of amniote evolution, Plos One, 2, e889, https://doi.org/10.1371/journal.pone.0000889, 2007.
Oelofsen, B. and Araújo, D. C.: Paleoecological implications of the distribution of mesosaurid reptiles in the Permian Irati Sea (Parana Basin), South America, Revista Brasileira de Geociências, 13, 1–6, 1983.
Oelofsen, B. W. and Araújo, D. C.: Mesosaurus tenuidens and Stereosternum tumidum from the Permian Gondwana of both Southern Africa and South America, S. Afr. J. Sci., 83, 370–372, 1987.
Olson, E. C.: The family Diadectidae and its bearing on the classification of reptiles, Chicago Natural History Museum, 1947.
Piñeiro, G., Ferigolo, J., Meneghel, M., and Laurin, M.: The oldest known amniotic embryos suggest viviparity in mesosaurs, Hist. Biol., 24, 620–630, 2012a.
Piñeiro, G., Ramos, A., Goso, C., Scarabino, F., and Laurin, M.: Unusual environmental conditions preserve a Permian mesosaur-bearing Konservat-Lagerstätte from Uruguay, Acta Palaeontol. Pol., 57, 299–318, 2012b.
Reisz, R. R., Mueller, J., Tsuji, L., and Scott, D.: The cranial osteology of Belebey vegrandis (Parareptilia: Bolosauridae), from the Middle Permian of Russia, and its bearing on reptilian evolution, Zool. J. Linn. Soc., 151, 191–214, 2007.
Richardson, M. K.: Vertebrate evolution: the developmental origins of adult variation, BioEssays, 21, 604–613, 1999.
Richardson, M. K., Gobes, S. M. H., van Leuwen, A. C., Polman, J. A. E., Pieau, C., and Sánchez-Villagra, M. R.: Heterochrony in limb evolution: developmental mechanisms and natural selection, J. Exp. Zool., 312, 639–664, 2009.
Rieppel, O.: Studies on skeleton formation in reptiles. IV. The homology of the reptilian (Amniote) astragalus revisited, J. Vertebr. Paleontol., 13, 31–47, 1993.
Rossmann, T. and Maisch, M. W.: Das Mesosaurier-Material in der Bayerischen Staatssammlung für Palaontologie und Historische Geologie: Übersicht und neue Erkenntnisse, Mitteilungen der Bayerischen Staatssammlung für Palaontologie und historische Geologie, 39, 69–83, 1999.
Sánchez-Villagra, M. R.: Developmental palaeontology in synapsids: the fossil record of ontogeny in mammals and their closest relatives, P. Roy. Soc. Lond. B Bio., 277, 1139–1147, 2010.
Spencer, P. S. and Lee, M. S. Y.: A juvenile Elginia and early growth in pareiasaurs, J. Paleontol., 74, 1191–1195, 2000.
Szczygielski, T., Surmik, D., Kapuścińska, A., and Rothschild, B. M.: The oldest record of aquatic amniote congenital scoliosis, PloS one, 12, e0185338, https://doi.org/10.1371/journal.pone.0185338, 2017.
Tsuji, L. A.: Anatomy, cranial ontogeny and phylogenetic relationships of the pareiasaur Deltavjatia rossicus from the Late Permian of central Russia, Earth Env. Sci. T. R. So., 104, 1–42, 2013.
Tsuji, L. A. and Müller, J.: Assembling the history of the Parareptilia: phylogeny, diversification, and a new definition of the clade, Foss. Rec., 12, 71–81, https://doi.org/10.1002/mmng.200800011, 2009.
Villamil, J., Demarco, P. N., Meneghel, M., Blanco, R. E., Jones, W., Rinderknecht, A., Laurin, M., and Piñeiro, G.: Optimal swimming speed estimates in the Early Permian mesosaurid Mesosaurus tenuidens (Gervais 1865) from Uruguay, Hist. Biol., 28, 963–971, 2016.