the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
The new encrusting microproblematicum Circumpodium enigmaticum and its attachment trace Circumpodichnus serialis from the Middle Jurassic of Normandy (France)
Liane Hüne
A single specimen of an enigmatic new attachment etching, together with an unknown calcareous encruster partly preserved in situ, has been identified on a belemnite rostrum from the Marnes de Dives Formation (Callovian, Middle Jurassic) of the Falaises des Vaches Noires in Normandy, France. The trace fossil, here established as the new ichnotaxon Circumpodichnus serialis igen. et isp. n., is a uniserial arrangement of very shallow depressions, oval to fusiform in outline, with peripheral pouches and central pits.
The trace maker has a morphology unlike any other known calcareous epibiont, fossil or recent, and is consequently described as the new microproblematicum Circumpodium enigmaticum gen. et sp. n. Its calcitic skeleton is composed of a chain of segments with perforate basal and lateral walls, anchored to the attachment trace in the substratum by vertical protrusions in the centre and feet-like protrusions in the periphery. The hypothetical upper wall of the segments was either organic-walled and has decayed or it was calcitic and has been abraded.
Based on morphological criteria and the capacity to bioerode, C. enigmaticum can best be compared to encrusting bryozoans and foraminiferans. Candidate bryozoans are aberrant arachnidiid ctenostomes, early cheilostomes, or stomatoporid cyclostomes. Among the foraminiferans, webbinellid or ramulinid polymorphinids are closest in their characters. In addition, tintinnid or folliculinid ciliophorans are considered as an alternative interpretation, and similarities to the Palaeozoic microproblematicum Allonema are discussed.
It is only rarely the case that ichnology is blessed with trace fossils that are preserved with their trace makers in situ. Here, we document such a rare case, specifically an encrusting calcareous microfossil preserved on a belemnite rostrum from the Middle Jurassic of Normandy (northern France), showing a delicate attachment etching in the host substrate. The identity of the trace maker, though, is all but straight forward to reveal, given that its morphology appears unlike any other known calcareous encruster, fossil or recent.
Acquiring the opinions of leading experts on a number of suspected trace maker groups led to a ramifying chain and loops of doubt. Where the attribution to a certain group was not entirely rejected but tentatively confirmed, it was stressed that we would deal with a new species or unknown ancestor. In consequence, we are left with what appears to be a challenging microproblematicum, for which we have a suite of alternative interpretations to offer. These include various types of bryozoans and foraminiferans, the two most diverse groups of calcifying encrusters with ability to bioerode, but also interpretations not commonly considered in calcifying epibionts.
Despite its uncertain higher systematic affinity, the encruster is herein established as a new genus and species in the informal group of Microproblematica, employing universal terminology that is as neutral as possible. On this basis, we call for feedback from the biology and palaeobiology research communities, in the hope that the systematic and taxonomic classification can be resolved and the palaeobiology of the organism eventually considered in the context of the proper organism group.
The bioerosion trace fossil that is preserved together with the microfossil is new to science too and can thus be established as new ichnotaxon. This is because ichnotaxonomy is a concept that is independent of the trace maker's identity – and the present case underlines why. Revealing the identity of the trace maker would nevertheless be an asset for interpreting the observed bioerosion pattern, while the ichnological evidence, in turn, may help in unravelling the nature of the enigmatic encrusting microproblematicum.
The Falaises des Vaches Noires (meaning “cliffs of the black cows”) are a stretch of coastal cliffs along the English Channel, 5 km in length and up to 110 m tall, situated between Villers-sur-Mer and Houlgate in the department of Calvados in Normandy, northern France. Ever since the monk Jacques-François Dicquemare published his observations on fossil cephalopods (Dicquemare, 1776a) and bones (Dicquemare, 1776b), it has become a classical locality for collecting and studying Middle Jurassic (upper Callovian), Upper Jurassic (lower and middle Oxfordian), and Cretaceous (lower Cenomanian) fossils (Dugué et al., 1998; Merles, 2011; Lebrun and Courville, 2013).
The base of the cliffs is formed by the upper Callovian Marnes de Dives Formation that reaches far into the sea to form a tidal platform emerging during low tide. These fossiliferous strata are an alternation of marls and clays with intercalated layers rich in nodules (Dugué et al., 1998). Among the diverse invertebrate fossils is the dwarf belemnite Hibolithes girardoti (Loriol, 1902), one of which carries the new microfossil and attachment trace fossil. As a general observation, these belemnites exhibit only a few encrusters but a particularly rich assemblage of bioerosion trace fossils. The analysis of a total of 56 well-preserved such belemnite rostra has yielded 16 bioerosion ichnospecies and three unnamed traces of euendoliths, presumably produced by annelid and phoronid worms, bryozoans, foraminiferans, cirripeds, sponges, brachiopods, and micro-fungi (Hüne, 2019).
According to the analysis of Rioult et al. (1991) the sequence stratigraphy of the Marnes de Dives Formation indicates a regressive phase and sequence boundary at its base, followed by a transgressive system tract with a maximum flooding surface near the top, indicative of an outer shelf depositional setting with terrigenous input from the Armorican Massif in the southwest. The fossils reported herein were deposited in the distal part of the transgressive system tract during sea-level highstand, as they stem from the middle member sensu Rioult et al. (1991), respectively the horizons H1 to H3 sensu Hébert (1860), and Douvillé (1881). This member is 2.5 m thick and corresponds to the Henrici subzone in ammonite biostratigraphy (Rioult et al., 1991).
The belemnite with the encrusting microfossil was photographed with a Nikon D800 DSLR equipped with a Micro Nikkor 60 mm 1:2.8 G ED macro lens and extension tubes, applying a Cognisys StackShot Macro Rail for extended focal imaging with the software Helicon Focus Pro. Close-ups were photographed with a Keyence VHX-2000 digital microscope, applying automated extended focal imaging. Scanning electron micrographs (SEMs) of the traces were produced with a Tescan VEGA3 XMU using the backscatter electron detector (BSE) in low-vacuum mode at 10 and 20 keV, allowing visualization of the sample without prior sputter coating. Morphometrical measurements were done with the measurements tool in the VEGA SEM software.
For an elemental analysis of the encrusting microfossil, the SEM's Oxford X-Max energy dispersive X-ray spectrometer (EDS) with INCA software (v. 5.05) was used. Spectral analyses were carried out at 20 keV acceleration voltage by scanning an area of 25 µm×25 µm for 180 s, with process time three, 2000 channels, and a beam intensity of 15, resulting in > 1 000 000 counts for the spectra.
This published work and the nomenclatural acts it contains have been registered with ZooBank: http://zoobank.org/references/CB8DCEBB-6D43-41A3-8894-0655C361AB43 (25 July 2019).
The enigmatic microfossil is preserved near the tip of a 31 mm long belemnite rostrum of the species Hibolithes girardoti (Fig. 1). The microfossil forms a meandering chain of seven oval to pyriform segments (numbered no. 1 to 7 in Figs. 1–2 and 4), two of which are complete (no. 5 and 7) and another two of which are fragmented (no. 2 and 4). The complete arrangement is indicated by means of faint attachment etchings on the surface of the belemnite. The segments of the microfossil consist of a flat-bottomed skeletal structure with a ring-shaped outer wall and conspicuous ramifying “feet” around the circumference (Figs. 2–3). The latter partly extend into etched pouches framing the attachment trace. Vertical protrusions around the centre of the segments provide an additional anchor (Figs. 3–4).

Figure 1Holotype of the microproblematicum Circumpodium enigmaticum gen. et sp. n. and its attachment trace fossil Circumpodichnus serialis igen. et isp. n., preserved on a rostrum of the belemnite Hibolithes girardoti from the Marnes de Dives Formation (Callovian, Middle Jurassic) at the Falaises des Vaches Noires in Normandy, France (SMF XXX 905 a and b). (a) Belemnite in natural size (length = 31 mm). (b) Tip of the belemnite with numbered segments of the calcareous encruster (partly preserved only) and the faint attachment trace below. (c) Close-up of segment no. 7. (d) Close-up of segment no. 5 and connection to no. 4. (e) Close-up of feet-like peripheral protrusions.
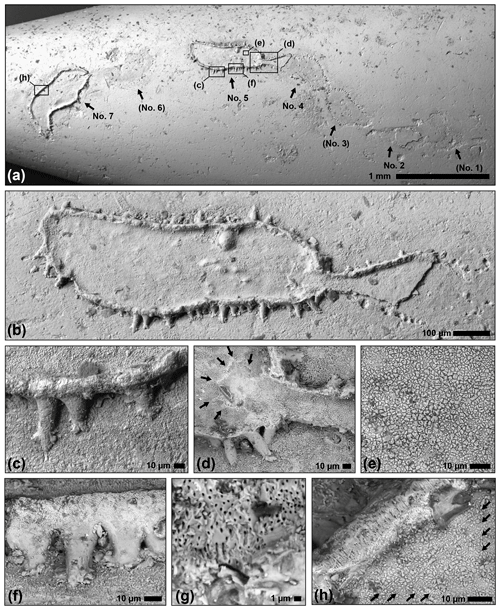
Figure 2Scanning electron micrographs (BSE detector) of the holotype of the microproblematicum Circumpodium enigmaticum gen. et sp. n. (SMF XXX 905 a). (a) Overview of the serial arrangement of segments (no. 1–7) with indication of close-ups; remains of the encruster are preserved in segments no. 2, 4, 5, and 7 only. (b) Segment no. 5 and the connection to segment no. 4. (c) Lateral wall with feet-like protrusions. (d) Stellate architecture (arrows) where the connection from the previous segment is anchored. (e) Polygonal calcite crystallites on the basal wall of the encruster. (f–g) Overview and close-up of the densely spaced minute pores that traverse the peripheral wall. (h) Faint striations in the basal wall (arrows), and the perpendicular pores traversing the peripheral wall of the encruster.
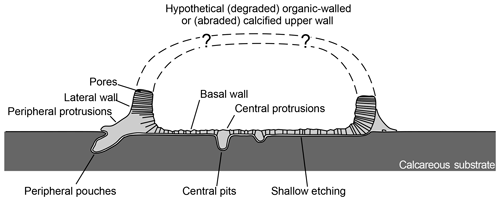
Figure 3Schematic transversal cross section of Circumpodium enigmaticum gen. et sp. n. and its attachment etching Circumpodichnus serialis igen. et isp. n.

Figure 4Scanning electron micrographs (BSE detector) of the holotype of the attachment trace Circumpodichnus serialis igen. et isp. n. (SMF XXX 905 b). (a) Overview of the serial arrangement of segments (no. 1–7) with indication of close-ups. (b) Segment no. 3 showing the diagnostic shallow etching trace with a peripheral array of pouch-shaped etchings, complemented by vertical pits in the centre. In the upper left, a continuous connection to a neighbouring segment is illustrated, whereas in the lower right the chain of etchings is intermittent. (c) Close-up of peripheral pouches, radiating and inclined outwards. (d) Close-up of circular pits, developed vertically near the centre of the segments (protrusions of trace maker still inside).
Two elemental EDS spectra were acquired, one at the central basal wall of the microfossil and another one at one of the “feet”. Both spectra yielded an elemental composition primarily composed of calcium, carbon, and oxygen, with a stoichiometric relationship close to that of CaCO3, hence identifying the skeletal mineral as calcium carbonate. In both spectra, traces of magnesium were detected (0.45 mass % and 1.01 mass %), suggesting calcite as the carbonate phase, as aragonite usually is devoid of this trace element.
In the following account, both the calcareous microfossil and the attachment trace will be described in detail and taxonomically treated by applying zoological biotaxonomy and ichnotaxonomy, respectively.
-
Informal group of Microproblematica
-
Genus Circumpodium nov.
LSID: urn:lsid:zoobank.org:act:0E736BE2-D46E-
4394-A80B-0810A85ABB9E
Diagnosis
Calcareous encruster composed of serial chains of flat-bottomed segments with peripheral protrusions anchoring the structure to the substrate.
Type species
Circumpodium enigmaticum nov.
Etymology
Compound of the Latin circum (meaning in a circle, encircling, around) and the English “podium” (meaning foot or footstalk in anatomy and botany), borrowed from the Latin podium, which derives from the Ancient Greek pódion (πδιoν, meaning base), from the diminutive of poús (πoς, meaning foot); in reference to the peripheral arrangement of feet-like protrusions.
Remarks
While the microfossil bears affinity to some bryozoan and foraminiferan genera, its unique characters merit the establishment of a new genus. The morphological similarity to the microproblematicum Allonema Ulrich and Bassler, 1904 is discussed in the respective section below.
-
Species Circumpodium enigmaticum nov.
LSID: urn:lsid:zoobank.org:act:C5D0417C-A164-
4D3F-98B5-0E1375939C9CFigs. 1–3
Diagnosis
Segments oval, fusiform, or pyriform in outline, connected by a neck anchored within the subsequent segment, walls perforate, scattered central protrusions vertical, and densely spaced peripheral protrusions feet-shaped.
Etymology
Latinized from the English “enigmatic”, which derives from the Latin aenigmaticus and the Ancient Greek ainigma (α`′ινιγμα, meaning a riddle); in reference to the unresolved nature of the organism.
Type material, locality, and horizon
Holotype (Figs. 1–2) on a rostrum of the belemnite Hibolithes girardoti extracted from the 2.5 m thick middle member (Henrici subzone) of the Marnes de Dives Formation, Callovian, Middle Jurassic; sampled at the Falaises des Vaches Noires between Villers-sur-Mer and Houlgate, department of Calvados, Normandy, France. Deposited in the trace fossil collection of the Senckenberg Institute in Frankfurt, Germany (SMF XXX 905 a).
Description
The holotype of the calcareous encruster is comprised of a 5.5 mm long uniserial chain of seven segments, two of which are complete, two fragmented, and the outline of the remaining three only indirectly recorded by the attachment trace below (Figs. 1, 2a). The dimension of the oval, fusiform, or pyriform segments (partly derived from the attachment etchings) are 565 to 940 µm (mean: 721±120 µm) maximum length and 187 to 380 µm (mean: 281±59 µm) maximum width, with an overall increase in segment dimension along the chain (from segment no. 1 to 7). At the points of contact between the segments, the structure narrows to a neck, 45 to 86 µm (mean: 72±19 µm) wide. Each segment is composed of a calcified basal wall, about 5 to 8 µm in thickness, surrounded by a vertical rim, 7 to 13 µm in width and up to 40 µm in relatively constant height (Figs. 1c–d, 2a–b; schematic cross section illustrated in Fig. 3). From this rim, peripheral feet-like protrusions emerge, about 50 to 75 in number per segment, up to 55 µm long and 25 µm wide each, partly ramifying into short toe-like structures at the contact with the substrate surface (Figs. 1e, 2c–d). The neck extends into the proceeding segment, where it is anchored by a stellate structure (Fig. 2d). This structure lends the segments a polarity.
Skeletal microstructure
The skeleton appears mainly composed of fibrous calcite crystallites or polygonal prisms, 1 to 3 µm in diameter, that are best visible on the basal wall of the segments (Fig. 2e). In some places, faint striations, parallel to the outer rim, are developed (Fig. 2h). The outer rim and less pronounced the basal wall are perforated by minute pores, circular and straight, 0.2 to 0.4 µm in diameter, traversing the wall perpendicularly (Fig. 2f–h). The density of these pores appears to increase towards the top of the peripheral rim, whereas the feet-like protrusions have a smooth surface devoid of pores (Fig. 2f). The presence or absence of a central canal in the protrusions could not be verified, but at least there are no points of entry to such a structure visible on the inner side of the wall.
Remarks
The microfossil features an attachment etching in the host substrate that is not part of the diagnosis. Instead, this trace fossil is established as new ichnotaxon below.
-
Ichnofamily Podichnidae Wisshak, Knaust and
Bertling, 2019 -
Ichnogenus Circumpodichnus nov.
LSID: urn:lsid:zoobank.org:act:C9B1FC9F-7AD9-
4ECB-9A36-67F89800FEFC
Diagnosis
Shallow attachment etchings with peripheral pouches.
Type ichnospecies
Circumpodichnus serialis nov.
Etymology
Compound of the Latin circum (meaning in a circle, encircling, around) and the ichnogenus Podichnus; in reference to the peripheral arrangement of Podichnus-like radiating etching pits.
Remarks
Traces of the new ichnogenus differ from all previously described attachment etchings. While the individual pits show some resemblance to brachiopod attachment traces in the ichnogenus Podichnus Bromley and Surlyk, 1973, the spatial arrangement of these pits is very different from the dense centrifugal clusters exhibited by these ichnospecies. Individual ring-shaped diatom attachment etchings of the ichnogenus Ophthalmichnus Wisshak et al., 2014 differ by the ring being a continuous groove that is not composed of individual pits. The same applies to the more complex Augoichnus Arendt, 2012, an attachment trace of unknown trace makers (possibly gastropods) that may occur in serial arrangement. Another attachment trace with uniserial arrangement is found in the ichnogenus Finichnus Taylor et al., 2013, specifically the ichnospecies F. dromeus (Taylor et al., 1999). These etchings of cheilostome bryozoans show oval depressions only. Finally, the uniserial foraminiferan attachment traces of the ichnogenus Camarichnus Santos and Mayoral, 2006 differ in that they are composed of very shallow grooves that are connected by a central furrow.
-
Ichnospecies Circumpodichnus serialis nov.
LSID: urn:lsid:zoobank.org:act:521E7BD3-973E-
45E5-8E48-D04050FAE1EAFigs. 1, 3–4
Diagnosis
Serial arrangements of connected or intermittent shallow etchings, each oval, fusiform, or pyriform in outline, featuring densely spaced, radially inclined, lobed pouches in the periphery and sporadic, vertical, circular pits in the centre.
Etymology
Latinized from the English “serial”, which derives from the Latin series (meaning array, chain, string); in reference to the serial arrangement of the trace.
Type material, locality, and horizon
Holotype (Figs. 1, 4) on a rostrum of the belemnite Hibolithes girardoti extracted from the 2.5 m thick middle member (Henrici subzone) of the Marnes de Dives Formation, Callovian, Middle Jurassic; sampled at the Falaises des Vaches Noires between Villers-sur-Mer and Houlgate, department of Calvados, Normandy, France. Deposited in the trace fossil collection of the Senckenberg Institute in Frankfurt, Germany (SMF XXX 905 b).
Description
The holotype trace is a 5.5 mm long uniserial chain of seven attachment etchings, each 565 to 940 µm (mean: 721±120 µm) in maximum length and 187 to 380 µm (mean: 281±59 µm) in maximum width, with an overall increase in segment dimension along the chain (from segment no. 1 to 7). Five of these segments are connected by a constriction, 45 to 86 µm (mean: 72±19 µm) wide, whereas two are intermittent but in line and in close proximity (Figs. 1, 4a). Each segment is composed of an oval to pyriform, very shallow etching with 50 to 75 pouch-shaped pits in the periphery, radially inclined outwards (Fig. 4b; schematic cross section illustrated in Fig. 3). The opening of these pouches is about 15 to 40 long and 10 to 25 µm wide and they vary in depth (Fig. 4c). Around the centre of the segments there are about a dozen additional pits, vertical in orientation and circular in outline, with a diameter of about 10 to 15 µm (Fig. 4d).
Remarks
The trace maker is partly in place but explicitly not part of the trace fossil diagnosis.
6.1 Calcification
The perfect preservation of open pores in a submicron scale strongly suggests that the calcitic skeletal microstructure is reflecting the original mineralogy and structure. It can be assumed that pseudomorphosis of an organic skeletal substance, or recrystallization from aragonite, would have overprinted such delicate detail. Hence, we consider the microproblematicum in question to be a calcitic calcifier.
Whether the recorded segments are complete is a puzzling question, leaving room for speculation (Fig. 3). Possibly, only the basal portion of the chambers/zooecia/cells is preserved. In that case, the upper part could be missing due to abrasion. However, the relatively constant height of the rim might point to a different scenario. Could the upper portion of the skeleton have been cuticular and organic-walled whereas only the bottom part was calcified? With the present material we cannot answer this question, but in case uncalcified skeletal parts were involved, bioimmuration could shed light on the complete morphology of this microproblematicum. For the time being, any postulation upon the presence or absence of a larger aperture (other than the observed minute pores), which could have allowed a feeding device to protrude, would be entirely conjectural.
6.2 Bioerosion
It cannot entirely be excluded that pressure dissolution has created or partly enforced the observed pattern. However, it is again the perfect preservation of the pores that strongly suggests that pressure dissolution did not take place, as it would have overprinted such fine detail. Unless proven otherwise, the attachment structure reported herein is thus considered a bioerosion phenomenon.
Attachment etchings are a common category of bioerosion trace fossils, with currently 27 ichnospecies grouped in 17 ichnogenera in the ichnofamilies Podichnidae Wisshak et al., 2019; Renichnidae Wisshak et al., 2019; and Centrichnidae Wisshak et al., 2019. Based on the recorded behavioural pattern, they are categorized in the ethological class fixichnia (Gibert et al., 2004). They are produced by cheilostome bryozoans, brachiopods, vermetid and eulemid gastropods, anomiid bivalves, verrucid and balanid cirripeds, foraminiferans, and diatoms. These organisms presumably form the attachment etchings in order to enhance adhesion, offering better protection from hydrodynamics, grazers, and predators (for a review, see Bromley and Heinberg, 2006). With all likelihood, Circumpodium enigmaticum produced attachment etchings for the same reasons.
We assume that Circumpodichnus serialis was produced by means of chemical bioerosion, aided by the micropores in the basal and lateral walls of Circumpodium enigmaticum. These could have functioned in guiding chemical agents that lowered local pH and dissolved the calcareous substrate. We found no evidence for a central canal in the protrusions that could have alternatively served this purpose, but we cannot exclude that such a structure exists.
6.3 A bryozoan identity?
Candidates for the biological identity of Circumpodium enigmaticum are found among the bryozoans, the most diverse encrusting epibionts in many marine hard-bottom communities, past and present. The Middle Jurassic was a time of diversification and innovation in the phylum Bryozoa (Taylor, 1990a; Taylor and Ernst, 2008). Several principal groups of bryozoans deserve a closer consideration, in which case the observed segments would represent the individual zooecia and the serial arrangement would correspond to a runner-type, uniserial colony. The zooids, growing larger along the chain, would have been connected by stoloniferous necks.
Firstly, epilithic ctenostomes of the family Arachnidiidae Hincks, 1877 were abundant in the Jurassic, where they commonly formed uniserial chains of zooids. These zooids are in the same size range as C. enigmaticum, and they share the fusiform to pyriform outline. Some arachnidiids possess lateral attaching crenellations with some resemblance to the feet-like protrusions of the new microproblematicum, such as the Upper Jurassic Arachnoidella abusensis Taylor, 1990a or the extant Arachnidium lacourti d'Hondt and Faasse, 2006 and Arachnidium fibrosum Hincks, 1877. However, ctenostome bryozoans are – by definition – uncalcified, even though “crystalline structures” of an unspecified substance were reported for Arachnoidea dhondti Franzén and Sandberg, 2001 and believed by these authors to be homologous with the crenellations in other arachnidiid species. The fossil record of arachnidiids is sparse for reasons of their lack of calcification. In the Callovian Marnes de Dives Formation, they are recorded on belemnite rostra by means of epibiont shadowing (Fig. 5a–b). While these exhibit a similar outline to C. enigmaticum, they are not bioerosion structures but slightly elevated above the adjacent substrate surface (e.g. Palmer et al., 1993; Wilson and Taylor, 2012). Another mode of preservation is that of bioimmuration. That is, the soft bodied arachnidiids were overgrown by a skeletal encruster, such as the fixed valve of an oyster, preserving a negative imprint of the outer morphology (e.g. Taylor, 1990b; Todd, 1994). A derivation of this mode of preservation is cast bioimmuration, such as those of Arachnidium smithii (Phillips, 1829) recorded on a Hibolithes cf. semihastatus rotundus belemnite rostrum from the type locality (Fig. 5c–d). These natural casts (composed of calcite spar) of the internal structure of arachnidiid zooids had formed below bioimmurations – a process aptly addressed as “sandwiched fossils” by Taylor and Todd (1990). In the Ctenostomata, bioerosion is a widespread phenomenon, with most species not representing epibionts but euendoliths (e.g. Pohowsky, 1978). For the epilithic arachnidiids, the formation of attachment etchings has not been reported, but such a habit has been documented for another encrusting ctenostome bryozoan – the Pliocene Paravinella sekei Mayoral, 1987 that produced the stellate trace fossil Stellichnus radiatus Mayoral, 1987. Together with the morphological similarities outlined above, this suggests some possibility that C. enigmaticum represents an aberrant, i.e. at least partly calcified, epilithic ctenostome bryozoan with affinity to the Arachnidiidae. However, the ability to calcify would need to be explained. Also, a more recurrent outline of the zooids and distolateral branching would be expected.

Figure 5Records of arachnidiid bryozoans in the Marnes de Dives Formation (Callovian, Middle Jurassic) at the Falaises des Vaches Noires (Normandy, France). (a–b) Overview and SEM close-up of epibiont shadows left behind by an arachnidiid on a Hibolithes girardoti belemnite rostrum. (c–d) Overview and SEM close-up of cast bioimmurations of the arachnidiid species Arachnidium smithii, developed below a bioimmuration by a fixed oyster valve on a Hibolithes rostrum.
The prerequisite of the ability to calcify leads us to another group of bryozoans – the early cheilostomes. They have calcified zooid body walls, and Jurassic species often developed uniserial chains of zooecia in the same size range and outline as C. enigmaticum. Their primitive zooidal morphology is characterized by oval to pyriform autozooids having elliptical opesia surrounded by mural rims (Ostrovsky et al., 2008). The ability of cheilostomes to produce shallow attachment etchings (ichnogenera Finichnus Taylor et al., 2013 and Stellichnus Mayoral, 1987) is well established, including uniserial patterns, as represented by F. dromeus (Taylor et al., 1999). However, they neither show the peripheral pouches or central pits of Circumpodichnus serialis nor are feet-like protrusions known from the early cheilostomes. Calcified, basal attachment structures do exist in more advanced species and may readily fossilize (see e.g. Berning, 2006, figs. 23–24), although these are not known to penetrate/etch the substratum. Based on the scarce fossil record, the cheilostomes are considered to not have evolved before the Upper Jurassic (Oxfordian or Kimmeridgian; Taylor, 1994; Ostrovsky et al., 2008). They are believed to have evolved from arachnidiid ctenostomes after having developed the ability to calcify the zooid body walls and to form an operculum (Taylor, 1990a; Nikulina, 2002). This raises the question of whether C. enigmaticum could represent a previously unknown protocheilostome that had evolved already in the uppermost Middle Jurassic. Counterarguments would again include the little recurrent shape of the zooids.
A third group of bryozoans of relevance are stomatoporid cyclostomes. In contrast to ctenostomes and cheilostomes, their zooids are less recurrent in outline and thus more in accordance with the observed variability in C. enigmaticum. They can build uniserial runner-type colonies with fusiform to pyriform zooecia and were around already in the Middle Jurassic (e.g. Wilson et al., 2015; Zatoń and Taylor, 2009, 2010), with a peak in diversity in the Callovian (Zatoń et al., 2013). Their calcified zooid walls possess densely spaced pseudopores that might correspond to the densely spaced micropores observed in C. enigmaticum. Lateral protrusions are unknown from these cyclostomes, and the constriction between the individual zooecia is usually wider than the necks in C. enigmaticum. The ability to etch attachment traces has not been reported for cyclostomes. Nevertheless, at present, we cannot rule out a cyclostome identity for C. enigmaticum.
6.4 A foraminiferan identity?
The second obvious candidates are adherent foraminiferans, in which case the observed segments would represent interconnected chambers of an elongate calcareous foraminiferan, and the perforation with micropores would have served fluid exchange with the ambience or allowed pseudopodia to protrude. The chambers would have grown larger along the chain, with each chamber overgrowing the stoloniferous connection from the previous chamber. The somewhat contorted outline of segment no. 1 may or may not have carried an initial spiral.
The ability to bioerode is well documented for many foraminiferans (for reviews, see Vénec-Peyré, 1996, and Walker et al., 2017). This includes both the formation of attachment etchings (e.g. the ichnogenera Camarichnus Santos and Mayoral, 2006; Canalichnus Santos and Mayoral, 2006; and Kardopomorphos Beuck et al., 2008) as well as deeper, dendritic borings, presumably produced by naked euendolithic foraminiferans (e.g. the ichnogenus Nododendrina Vogel et al., 1987; see discussion in Bromley et al., 2007, and review in Wisshak, 2017).
There is some superficial similarity of C. enigmaticum to the nubeculariid Vinelloidea crussolensis Canu, 1913 (which is a senior synonym of Nubeculinella bigoti Cushman, 1930; see discussion in Voigt, 1973), which was originally described as a bryozoan and is a very common encrusting foraminiferan co-occurring with C. enigmaticum in the Marnes de Dives Formation (Fig. 6a). However, V. crussolensis does not etch an attachment trace, and SEM images furthermore show that it lacks lateral protrusions and has a different microstructure (Fig. 6b). However, Vinelloidea and related genera in the family Nubeculariidae Jones in Griffith and Henfrey, 1875 are porcelaneous, constructed of randomly oriented rod-like crystals, and imperforate (Loeblich and Tappan, 1987). We illustrate V. crussolensis here to document these marked differences.

Figure 6Encrusting foraminiferans from the Marnes de Dives Formation (Callovian, Middle Jurassic) at the Falaises des Vaches Noires (Normandy, France). (a) Vinelloidea crussolensis on an oyster valve, with surficial similarity to C. enigmaticum in its meandering and multicamerate mode of encrustation. (b) SEM close-up of partly eroded chamber, illustrating marked differences in the microstructure and lack of attachment etching below the partly detached lateral wall. (c) Bullopora aff. rostrata on a bivalve shell. (d–e) SEM images of the same specimen, illustrating a slight tuberculation, a continuous fringe, and the perforate microstructure.
Instead, circumstantial evidence points towards a different group, namely fistulose foraminiferans of the family Polymorphinidae d'Orbigny, 1839. They attach themselves to hard substrates via lateral branching appendages somewhat similar to the feet-like protrusions of C. enigmaticum (e.g. Pożaryska and Voigt, 1985; Guilbault et al., 2006). These projections contain tubes leading to the fistulose apertures at their tips, but the presence of such internal structures could not be confirmed, albeit not entirely rejected, for C. enigmaticum. The Polymorphinidae include multichambered forms attached to the substrate primarily by the outer wall of their tests, with chambers loosely connected by stolon-like necks (Loeblich and Tappan, 1987). Foremost, these are species in the subfamilies Webbinellinae Rhumbler, 1904 and Ramulininae Brady, 1884. The former subfamily includes, for instance, the genus Bullopora Quenstedt, 1856, whose species are very common encrusters on Jurassic and Cretaceous belemnite rostra and other substrates (e.g. Adams, 1962; Pugaczewska, 1965; Hart et al., 2009) and who show a similarity in segmentation to C. enigmaticum. A specimen from the Marnes de Dives Formation is illustrated in Fig. 6c–d. None of the Bullopora species were diagnosed with feet-like protrusions though, but at least one has a pronounced tuberculation – Bullopora tuberculata Sollas, 1877, first described from the Late Cretaceous (Cenomanian) Cambridge Greensand. Such tuberculation has also been reported for some specimens of the type species Bullopora rostrata Quenstedt, 1857 from the Upper Jurassic Oxford and Kimmeridge Clay, leading Adams (1962) to propose B. rostrata var. irregularis, for which he also noted that “an irregular basal flange formed of finger-like calcareous processes from the wall is often present”. Such ornamented forms of Bullopora are more common in the Upper Jurassic and particularly the Cretaceous (see discussion in Barnard, 1958), where they show affinities for the more heavily spined representatives of the genus Ramulina Jones in Wright, 1875. All these polymorphinids have calcitic skeletons with walls perforated by minute pores (see also Fig. 6e), which is in good accordance with the skeletal composition and microstructure of C. enigmaticum. According to Loeblich and Tappan, 1987, the Polymorphinidae first appear in the Upper Triassic, with the record of the Webbinellinae and Ramulininae starting in the Jurassic.
A bioeroding habit was documented for the Eocene to Pliocene, ectoparasitic, polymorphinid Vasiglobulina alabamensis (Cushman and McGlamery, 1939) whose spines penetrate shell material, leaving small round traces (Poag, 1969; Walker, 2017). Furthermore, according to Walker (2017) some species of the aforementioned genus Ramulina are suspected endoparasites and bioerode excavations that match the size of their chambers, or they produce small round holes that penetrate the host skeleton. In particular, these are the Late Cretaceous to extant R. globulifera (Brady, 1879) and the Oligocene R. parasitica Carter, 1889.
Together these characteristics of the Polymorphinidae, especially the subfamilies Webbinellinae and Ramulininae, render these adherent foraminiferans perhaps the most promising direction for the identity of C. enigmaticum.
6.5 A ciliophoran identity?
Yet another possibility to explore, albeit rarely considered as calcifying epibionts, is ciliate protozoa (Ciliophora). In this case, the segments of C. enigmaticum would correspond to linked unicellular sessile ciliophorans, with a calcified base of the lorica (ciliophoran test) attached to the hard substrate.
Most ciliophorans are organic-walled only, but for several species, particularly among the order of the Tintinnida Kofoid and Campbell, 1929, the ability to calcify the lorica was reported (Corliss, 1979; Armstrong and Brasier, 2005). Some of these are Jurassic genera of the family Calpionellidae Bonet, 1956, a group of microfossils whose affinity to the tintinnids bears considerable doubt because their composition differs from all known extant tintinnids (see discussion in Lipps et al., 2013). Another family to consider are the Folliculinidae Dons, 1914 in the order Heterotrichida Stein, 1859. Folliculinid ciliates have a lorica that they attach to a hard surface and an aperture through which cilia apparatuses extend (e.g. Andrews, 1914; Das, 1949; Hadži, 1951).
Due to the poor fossilization potential of these delicate and mostly organic-walled structures, the fossil record of the tintinnids and folliculinids is very scarce. The earliest known unequivocal ciliate fossil tintinnids occur in the Jurassic, even though numerous putative tintinnids were reported also from Palaeozoic and Proterozoic strata (for reviews, see Tappan and Loeblich, 1968, and Lipps et al., 2013). The fossil record of the folliculinids is unreliable but can be expected to reach back into the Mesozoic (Jere Lipps, personal communication, 2019).
While it is conceivable that C. enigmaticum represents the partly calcified lorica of such ciliophorans, what remains puzzling is their uniserial arrangement. According to Corliss (1979), the lorica of some ciliophorans may occur in a multiple arboroid-tree state that might explain this observed pattern. Frontal budding at their base could perhaps be an underlying process to form such a chain.
6.6 Affinity to the Palaeozoic microproblematicum Allonema
Circumpodium enigmaticum exhibits considerable similarity to another puzzling microproblematicum, namely Allonema Ulrich and Bassler, 1904, both in terms of morphology as well as interpretation. The various species of this incertae sedis (1) also occur in uniserial chains of bead-shaped segments, (2) are in the same size range (about 200 to 1000 µm in length and 100 to 200 µm in width), (3) also have planar junctions with adjacent vesicles, (4) have a skeleton riddled with pores (albeit larger in diameter; 2 to 20 µm), and (5) are also composed of calcite (Wilson and Taylor, 2014). Allonema differs in that the individual vesicles do not show a polarity and by a lack of feet-like protrusions. Furthermore, Allonema shows a morphological continuum to the related microproblematicum Ascodictyon Nicholson and Etheridge, 1877 with features not observed in C. enigmaticum, such as longer stolons or stellate aggregates. For these reasons, assignation of the new microproblematicum to Allonema is deemed unfeasible. Finally, there is a significant discrepancy in abundance and in stratigraphic range, with Allonema being widespread and restricted to the Palaeozoic (Ordovician to Permian; Jarochowska et al., 2016), respectively.
Notwithstanding, the given similarities deserve attention. Tellingly the interpretations that have been put forward for the Ascodictyon/Allonema complex (sensu Wilson and Taylor, 2014) explored similar directions by considering bryozoans and foraminiferans. While early workers refrained from deciding upon the biological identity (for a review, see Wilson and Taylor, 2014), a tentative assignment to the ctenostome bryozoans by Ulrich and Bassler (1904) had manifested for more than a century, leading Wilson and Taylor (2001, 2014) and others to revisit the species and to remove what they addressed in 2001 as “pseudobryozoans” from the Bryozoa. Instead they have postulated that Allonema/Ascodictyon vesicles most likely represent the encrusting base, possibly functioning as nutrient storage structures, of an organism with unknown erect parts that carry the feeding organs. In analogy to recent encrusters, this could still encompass bryozoans, similar to species in the extant genera Caulibugula Verrill, 1900 or Crisia Lamouroux, 1812. As an alternative interpretation, Wilson and Taylor (2014) underline the similarity to some encrusting foraminiferans.
The new microproblematicum Circumpodium enigmaticum gen. et sp. n. is a calcitic encruster composed of serial chains of segments with a calcified and perforate basal and lateral wall, anchored to the substratum by vertical protrusions in the centre and feet-like protrusions in the periphery. The hypothetical upper wall of the segments was either organic and has decayed or it was calcitic and has been abraded.
Based on morphological characters and the bioerosion capacity, C. enigmaticum can best be compared to encrusting bryozoans and foraminiferans. Additionally, it is compared to the microproblematicum Allonema, for which the same two affinities have been proposed. Among the bryozoans, candidate identities include aberrant arachnidiid ctenostomes, early cheilostomes, and stomatoporid cyclostomes. In the foraminiferans, webbinellid or ramulinid polymorphinids encompass the closest match. Apart from these obvious candidate organism groups, tintinnid or folliculinid ciliophorans are an alternative interpretation. More material with complete preservation (or bioimmuration) is needed to gain a better understanding of the morphology of C. enigmaticum and to allow evaluation of the presence and nature of an upper wall, the development of an orificium and operculum, and the presence/type of branching.
The shallow attachment etching with peripheral pouches and central pits is established as the new ichnotaxon Circumpodichnus serialis igen. et isp. n. It confirms the ability of C. enigmaticum to bioerode such a structure, presumably by chemical means, and probably to improve adhesion to the substrate. Recognition of further occurrences of this trace fossil may help in deciphering the stratigraphical range and preferred palaeoenvironment of the delicate encrusting microfossil. For the time being, the biological identity of C. enigmaticum remains a conundrum.
The type material is reposited in the Senckenberg collection (as specified in Sect. 5, Systematic palaeontology), and all relevant data are presented in this paper.
LH discovered and provided the belemnite carrying the two holotypes and additional material for comparison. MW studied these specimens in detail and prepared the manuscript with contributions from the co-author.
The authors declare that they have no conflict of interest.
We'd like to acknowledge the numerous experts, in various fields of (palaeo)biology research, who shared their opinions and suspicions in regard to the potential identity of the new microproblematicum. In alphabetical order, these were Sabine Agatha (Salzburg, Austria), Björn Berning (Linz, Austria), Markus Bertling (Münster, Germany), Gérard Breton (Le Havre, France), Micah Dunthorn (Essen, Germany), Peter Girod (Berlin, Germany), Michael Hesemann (Hamburg, Germany), Christian Klug (Zürich, Switzerland), Dirk Knaust (Stavanger, Norway), Jere Lipps (Berkeley, USA), Andres Rüggeberg (Fribourg, Switzerland), Joachim Schönfeld (Kiel, Germany), Paul Taylor (London, UK), and Mark Wilson (Wooster, USA). Finally, we thank Jere Lipps and Mark Wilson for their positive reviews and valuable suggestions.
This paper was edited by Rene Hoffmann and reviewed by Mark Wilson and Jere Lipps.
Adams, C. G.: Calcareous adherent foraminifera from the British Jurassic and Cretaceous and the French Eocene, Palaeontology, 5, 149–170, 1962.
Andrews, E. A.: The bottle-animalcule, Folliculina; œcological notes, Biol. Bull., 26, 262–285, 1914.
Arendt, Y. A.: Traces of Augoichnus dituberculatus gen. et sp. nov. on Hypermorphocrinus magnospinosus from the Lower Permian of Cisuralia, Paleontol. J., 46, 886–893, 2012.
Armstrong, H. A. and Brasier, M. D.: Microfossils, 2nd Edn., Blackwell Publishing, Malden, 296 pp., 2005.
Barnard, T.: Some Mesozoic adherent foraminifera, Palaeontology, 1, 116–124, 1958.
Berning, B.: The cheilostome bryozoan fauna from the Late Miocene of Niebla (Guadalquivir Basin, SW Spain): environmental and biogeographic implications, Mit. Geol.-Paläontol. Inst. Univ. Hamburg, 90, 7–156, 2006.
Beuck, L., López Correa, M., and Freiwald, A.: Biogeographical distribution of Hyrrokkin (Rosalinidae, Foraminifera) and its host-specific morphological and textural trace variability, in: Current developments in bioerosion, edited by: Wisshak, M. and Tapanila, L., Springer, Berlin, Heidelberg, 329–360, 2008.
Bonet, F.: Zonificación microfaunística de las calizas cretácicas del este de México, Contribución de la Asociación Mexicana de Geólogos Petroleros, 8, 413–488, 1956.
Brady, H. B.: Notes on some of the reticularian Rhizopoda of the “Challenger” Expedition; Part I. On new or little known arenaceous types, Quart. J. Microsc. Sci., 19, 20–67, 1879.
Brady, H. B.: Report on the Foraminifera dredged by H. M. S. Challenger, during the years 1873–1876, Longmans & Co, London, 814 pp., 1884.
Bromley, R. G. and Heinberg, C.: Attachment strategies of organisms on hard substrates: A palaeontological view, Palaeogeogr. Palaeocl., 232, 429–453, 2006.
Bromley, R. G. and Surlyk, F.: Borings produced by brachiopod pedicles, fossil and recent, Lethaia, 6, 349–365, 1973.
Bromley, R. G., Wisshak, M., Glaub, I., and Botquelen, A.: Ichnotaxonomic review of dendriniform borings attributed to foraminiferans: Semidendrina igen. nov., in: Trace fossils: concepts, problems, prospects, edited by: Miller III, W., Elsevier, Amsterdam, 518–530, 2007.
Canu, F.: Contributions a l'étude des Bryozoaires fossiles. Bryozoaires jurassiques, B. Soc. Géol. Fr., 13, 267–276, 1913.
Carter, H. J.: Ramulina parasitica, a new species of fossil foraminifera infesting Orbitolites mantelli, var. theobaldi, with comparative observations on the process of reproduction in the Mycetozoa, freshwater rhizopoda, and foraminifera, J. Nat. Hist., 4, 94–101, 1889.
Corliss, J. O.: The ciliated Protozoa: characterization, classification and guide to the literature, Pergamon Press, Oxford, 1979.
Cushman, J. A.: Note sur quelques foraminifères jurassiques d'Auberville (Calvados), Bulletin de la Société Linnéenne de Normandie, 8, 132–135, 1930.
Cushman, J. A. and McGlamery, W.: New species of Foraminifera from the Lower Oligocene of Alabama, Contributions from the Cushman Laboratory for Foraminiferal Research, 15, 45–49, 1939.
Das, S. M.: British Folliculinidae (Ciliata, Heterotricha), J. Mar. Biol. Assoc. UK, 28, 381–93, 1949.
d'Hondt, J.-L. and Faasse, M. A.: A new Arachnidium species, Arachnidium lacourti spec. nov. (Bryozoa: Ctenostomona), from the coast of The Netherlands, Zoologische Mededelingen, 80, 87–90, 2006.
Dicquemare, J.-F.: Suite des observations sur la nature & l'origine des coquilles fossiles, Observations sur la Physique, sur l'Histoire Naturelle et les Arts, 7, 38–41, 1776a.
Dicquemare, J.-F.: Ostéolithes, Observations sur la Physique, sur l'Histoire Naturelle et les Arts, 7, 406–414, 1776b.
Dons, C.: Folliculina-Studien IV. Vorläufige Bemerkungen über die Systematik der Folliculiniden nebst Beschreibung neuer norwegischer Arten, Tromsø Museums Arshefter, 35, 59–92, 1914.
d'Orbigny, A. D.: Foraminifères, in: Histoire physique, politique et naturelle de l'ile de Cuba, edited by: de la Sagra, R., Arthus Bertrand, Paris, 224 pp., 1839.
Douvillé, H.: Note sur la partie moyenne du terrain jurassique dans le bassin de Paris et sur le terrain coralline en particulier, B. Soc. Géol. Fr., 9, 439–474, 1881.
Dugué, O., Fily, G., and Rioult, M.: Le Jurassique des côtes du Calvados. Biostratigraphie, sédimentologie, paléoécologie, paléogéographie et stratigraphie séquentielle, Bulletin Trimestriel de la Société Géologique de Normandie et des Amis du Muséum du Havre, 85, 1–132, 1998.
Franzén, Å. and Sandberg, L.: Arachnoidea dhondti, a new species of bryozoan from the North Atlantic (Bryozoa, Ctenostomata), Cah. Biol. Mar., 42, 189–195, 2001.
Gibert, J. M., Domènech, R., and Martinell, J.: An ethological framework for animal bioerosion trace fossils upon mineral substrates with proposal of a new class, fixichnia, Lethaia, 37, 429–437, 2004.
Griffith, J. W. and Henfrey, A. (Eds.).: The micrographic dictionary: a guide to the examination and investigation of the structure and nature of microscopic objects, 3rd Edn., Van Voorst, London, 1060 pp., 1875.
Guilbault, J.-P., Krautter, M., Conway, K. W., and Barrie, J. V.: Modern foraminifera attached to Hexactinellid sponge meshwork on the West Canadian Shelf: Comparison with Jurassic Counterparts from Europe, Palaeontol. Electron., 9, 9.1.3A, 48 pp., available at: https://palaeo-electronica.org/2006_1/sponge/issue1_06.htm (last access: 8 October 2019), 2006.
Hadži, J.: Studien über Folliculiniden, Dela/Slovenska Akademija Znanosti in Umetnosti, 4, 1–350, 1951.
Hart, M. B., De Jonghe, A., Grimes, S. T., Metcalfe, B., Price, G. D., and Teece, C.: Microfaunal analysis of the Wattonensis Beds (Upper Bathonian) of South Dorset, Geosciences in South-West England, 12, 134–139, 2009.
Hébert, E.: Du terrain Jurassique supérieur sur les côtes de la Manche, B. Soc. Géol. Fr., 17, 300–316, 1860.
Hincks, T.: On British Polyzoa. Part I, Ann. Mag. Nat. Hist., 20, 212–218, 1877.
Hüne, L.: Traces de bioérosion sur des bélemnites jurassiques des Vaches Noires (Calvados, France), L'Écho des Falaises, Bulletin de l' Association Paléontologique de Villers-sur-Mer, 23, 41–54, 2019.
Jarochowska, E., Hierl, F., Vinn, O., and Munnecke, A.: Reducing taxonomic noise in problematic fossils: revision of the incertae sedis genus Allonema based on shape analysis, Bull. Geosci., 91, 97–110, 2016.
Kofoid, C. A. and Campbell, A. S.: A conspectus of the marine and freshwater Ciliata belonging to the suborder Tintinnoidea, with descriptions of new species principally from Agassiz expedition to the eastern tropical Pacific, 1904–1905, Univ. California Publ. Zool., 34, 1–403, 1929.
Lamouroux, J. V. F.: Extrait d'une mémoire sur la classification des Polypiers coralligènes non entièrement pierreux, N. Bull. Sci. Soc. Philosoph., 3, 181–188, 1812.
Lebrun, P. and Courville, P.: Le Jurassique des falaises des Vaches-Noires, Fossiles (Hors-Série), 4, 16–28, 2013.
Lipps, J. H., Stoeck, T., and Dunthorn, M.: Fossil tintinnids, in: The biology and ecology of tintinnid ciliates: models for marine plankton, edited by: Dolan, J. R., Montagnes, D. J. S., Agatha, S., Wayne Coats, D., and Stoecker, D. K., John Wiley & Sons, Oxford, 186–197, 2013.
Loeblich, A. R. and Tappan, H. N.: Foraminiferal genera and their classification, Van Nostrand Reinhold, New York, 1728 pp., 1987.
Loriol, P. de: Etude sur les mollusques et brachiopodes de I'Oxfordien supérieur et moyen du Jura lédonien, Mém. Soc. Paléontol. Suisse, 9, 1–76, 1902.
Mayoral, E.: Stellichnus nov. icnogen., huellas de incrustación atribuidas a Paravinella nov. gen. (Bryozoa, Ctenostomata) de la Formación Arenas de Huelva (Plioceno inferior) en la Cuenca del Bajo Guadalquivir (Espana), Rev. Esp. Paleontol., 2, 33–40, 1987.
Merles, B.: Les falaises des Vaches Noires de Cuvier au Paléospace, Éditions des Falaises, Rouen, 128 pp., 2011.
Nicholson, H. A. and Etheridge, R.: On Ascodictyon, a new provisional and anomalous genus of Palæozoic fossils, J. Nat. Hist., 19, 463–468, 1877.
Nikulina, E. A.: The evolution of colony morphogenesis in bryozoans of the order Cheilostomata, Paleontol. J., 36, 353–428, 2002.
Ostrovsky, A. N., Taylor, P. D., Dick, M. H., and Mawatari, S. F.: Pre-Cenomanian cheilostome Bryozoa: current state of knowledge, in: Proceedings of International Symposium The Origin and Evolution of Natural Diversity, 1–5 October 2007, Sapporo, Japan, 69–74, 2008.
Palmer, T. J., Taylor, P. D., and Todd, J. A.: Epibiont shadowing: a hitherto unrecognised way of preserving soft-bodied fossils, Terra Nova, 5, 568–572, 1993.
Phillips, J.: Illustrations of the Geology of Yorkshire, John Wiley & Sons, Oxford, 192 pp., 1829.
Poag, C. W.: Dissolution of molluscan calcite by the attached foraminifer Vasiglobulina, new genus (Vasiglobulininae, new subfamily), Tulane Stud. Geol. Paleontol., 7, 45–72, 1969.
Pohowsky, R. A.: The boring ctenostomate Bryozoa: taxonomy and paleobiology based on cavities in calcareous substrata, B. Am. Paleontol., 73, 1–192, 1978.
Pożaryska, K. and Voigt, E.: Bryozoans as substratum of fossil fistulose foraminifera (fam. Polymorphinidae), Lethaia, 18, 155–165, 1985.
Pugaczewska, H.: Les organismes sédentaires sur les rostres des bélemnites du Crétacé Supérieur, Acta Palaeontol. Pol., 10, 73–95, 1965.
Quenstedt, F. A.: Der Jura, Laupp, Tübingen, 842 + 224 pp., 1856–1858.
Rhumbler, L.: Systematische Zusammenstellung der recenten Reticulosa, Arch. Protistenkd., 3, 181–294, 1904.
Rioult, M., Dugué, O., Jan du Chêne, R., Ponsot, C., Fily, G., Moron, J.-M., and Vail, P. R.: Outcrop sequence stratigraphy of the Anglo-Paris Basin, Middle to Upper Jurassic (Normandy, Maine, Dorset), Bull. Cent. Rech. Expl., 15, 101–194, 1991.
Santos, A. and Mayoral, E.: Bioerosive structures of sclerozoan foraminifera from the lower Pliocene of southern Spain: a contribution to the palaeoecology of marine hard substrate communities, Palaeontology, 49, 719–732, 2006.
Sollas, W. J.: On the perforate character of the genus Webbina, with a notice of 2 new species, W. laevis and W. tuberculata, from the Cambridge Greensand, Geol. Mag., 4, 102–105, 1877.
Stein, F.: Der Organismus der Infusionsthiere nach eigenen Forschungen in systematischer Reihenfolge bearbeitet. I. Abtheilung. Allgemeiner Theil und Naturgeschichte der hypotrichen Infusionsthiere, Engelmann, Leipzig, 206 pp., 1859.
Tappan, H. and Loeblich, A. R.: Lorica composition of modern and fossil Tintinnida (ciliate Protozoa), systematics, geological distribution, and some new Tertiary taxa, J. Paleontol., 42, 1378–1394, 1968.
Taylor, P. D.: Bioimmured ctenostomes from the Jurassic and the origin of the cheilostome Bryozoa, Palaeontology, 33, 19–34, 1990a.
Taylor, P. D.: Preservation of soft-bodied and other organisms by bioimmuration – a review, Palaeontology, 33, 1–17, 1990b.
Taylor, P. D.: An early cheilostome bryozoan from the Upper Jurassic of Yemen, N. Jb. Geol. Paläontol. Abh., 191, 331–344, 1994.
Taylor, P. D. and Ernst, A.: Bryozoans in transition: the depauperate and patchy Jurassic biota, Palaeogeogr. Palaeocl., 263, 9–23, 2008.
Taylor, P. D. and Todd, J. A.: Sandwiched fossils, Geol. Today, September–October 1990, 151–154, 1990.
Taylor, P. D., Wilson, M. A., and Bromley, R. G.: A new ichnogenus for etchings made by cheilostome bryozoans into calcareous substrates, Palaeontology, 42, 595–604, 1999.
Taylor, P. D., Wilson, M. A., and Bromley, R. G.: Finichnus, a new name for the ichnogenus Leptichnus Taylor, Wilson and Bromley, 1999, preoccupied by Leptichnus Simroth, 1896 (Mollusca, Gastropoda), Palaeontology, 56, 456, 2013.
Todd, J. A.: The role of bioimmuration in the exceptional preservation of fossil ctenostomates, including a new Jurassic species of Buskia, in: Biology and palaeobiology of bryozoans, edited by: Hayward, P. J., Ryland, J. S., and Taylor, P. D., Olsen & Olsen, Fredensborg, 187–192, 1994.
Ulrich, E. O. and Bassler, R. S.: A revision of the Paleozoic Bryozoa, Smithsonian Misc. Coll., 45, 256–294, 1904.
Verrill, A. E.: Additions to the Tunicata and Molluscoidea of the Bermudas, Trans. Connecticut Acad. Art. Sci., 10, 592–594, 1900.
Vénec-Peyré, M.-T.: Bioeroding foraminifera: a review, Mar. Micropaleontol., 28, 19–30, 1996.
Vogel, K., Golubić, S., and Brett, C. E.: Endolith associations and their relation to facies distribution in the Middle Devonian of New York State, USA, Lethaia, 20, 263–290, 1987.
Voigt, E.: Vinelloidea CANU, 1913 (angeblich jurassische Bryozoa Ctenostomata) = Nubeculinella CUSHMAN, 1930 (Foraminifera). Paläont. Abh., 4, 665–670, 1973.
Walker, S. E., Hancock, L. G., and Bowser, S. S.: Diversity, biogeography, body size and fossil record of parasitic and suspected parasitic foraminifera: a review, J. Foramin. Res., 47, 34–55, 2017.
Wilson, M. A. and Taylor, P. D.: “Pseudobryozoans” and the problem of encruster diversity in the Paleozoic, PaleoBios, 21, 134–135, 2001.
Wilson, M. A. and Taylor, P. D.: Palaeoecology, preservation and taxonomy of encrusting ctenostome bryozoans inhabiting ammonite body chambers in the Late Cretaceous Pierre Shale of Wyoming and South Dakota, USA, in: Bryozoan studies 2010, edited by: Ernst, A., Schäfer, P., and Scholz, J., Springer, Berlin, Heidelberg, 419–433, 2012.
Wilson, M. A. and Taylor, P. D.: The morphology and affinities of Allonema and Ascodictyon, two abundant Palaeozoic encrusters commonly misattributed to the ctenostome bryozoans, Studi Trentini di Scienze Naturali, 94, 259–266, 2014.
Wilson, M. A., Bosch, S., and Taylor, P. D.: Middle Jurassic (Callovian) cyclostome bryozoans from the Tethyan tropics (Matmor Formation, southern Israel), Bull. Geosci., 90, 51–63, 2015.
Wisshak, M.: Taming an ichnotaxonomical Pandora's box: Revision of dendritic and rosetted microborings (ichnofamily: Dendrinidae), Eur. J. Taxon., 390, 1–99, 2017.
Wisshak, M., Alexandrakis, E., and Hoppenrath, M.: The diatom attachment scar Ophthalmichnus lyolithon igen. et isp. n., Ichnos, 21, 111–118, 2014.
Wisshak, M., Knaust, D., and Bertling, M.: Bioerosion ichnotaxa: review and annotated list, Facies, 65, 24, https://doi.org/10.1007/s10347-019-0561-8, 2019.
Wright, J.: A list of the Cretaceous microzoa of the north of Ireland. Proceedings Belfast Naturalists' Field Club (1873–1880), New Series, 1 (Appendix 31), 73–99, 1875.
Zatoń, M. and Taylor, P. D.: Middle Jurassic cyclostome bryozoans from the Polish Jura, Acta Palaeontol. Pol., 54, 267–288, 2009.
Zatoń, M. and Taylor, P. D.: Bathonian (Middle Jurassic) cyclostome bryozoans from the Polish Jura, Bull. Geosci., 85, 275–302, 2010.
Zatoń, M., Hara, U., Taylor, P. D., and Krobicki, M.: Callovian (Middle Jurassic) cyclostome bryozoans from the Zalas Quarry, southern Poland, Bull. Geosci., 88, 837–863, 2013.




