the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
An unfamiliar physeteroid periotic (Cetacea: Odontoceti) from the German middle–late Miocene North Sea basin at Groß Pampau
Irene Montañez-Rivera
Oliver Hampe
The Miocene mica clay locality of Groß Pampau, known for numerous and partly spectacular finds of marine mammals is becoming more and more a prominent site that bears the potential to resolve questions regarding taphonomic relationships and to interpret life communities of the ancient North Sea because of its rich faunal assemblage including invertebrates and other remains of various vertebrate organisms. In the present work we describe a right periotic of Physeteroidea with morphological characters so far unknown from other sperm whales. The periotics of the middle Miocene Aulophyseter morricei demonstrate the closest resemblance to the Groß Pampau specimen in their overall appearance and in the general arrangement and proportions of single structures, particularly of the anterior process and pars cochlearis. A great similarity is also documented with periotics of the living sperm whale, Physeter macrocephalus, especially regarding the shape and disposition of the anterior process and the bony element located dorsally to the accessory ossicle. Kogiid periotics differ strongly from that of the Groß Pampau specimen by having an inflated and short anterior process and, typically, three well-defined spines on it. A new taxonomic naming of the Groß Pampau periotic is not appropriate at this stage, although it might demonstrate the existence of a so-far undescribed physeteroid species. Additionally, its systematic position remains yet unclear and it is unknown at this point if it could belong to Hoplocetus ritzi, another physeterid, whose fragments were discovered in the same locality, or to another, already-described taxon, of which the periotic is still unknown.
A well-preserved but incomplete physeteroid periotic was recovered in May 2014 from the late Miocene mica clay deposits in a volume of clay of about 1 m3 in the eastern part of the commercial gravel pit of the Kieswerke Ohle & Lau company (dot number 8 in Fig. 1), located near the village Groß Pampau in Schleswig-Holstein, northern Germany. The periotic was found together with a part of a cranial bone (possibly a piece of a left paroccipital process of the exoccipital), a fragmentary bulla, about 25 small pieces of bone, one eroded centrum of a vertebra from the posteriormost vertebral column, and one distal phalanx. It remains unclear whether all discovered bone fragments belong to the same fossil specimen or not. They are too fragmentarily preserved to provide a sufficient description.
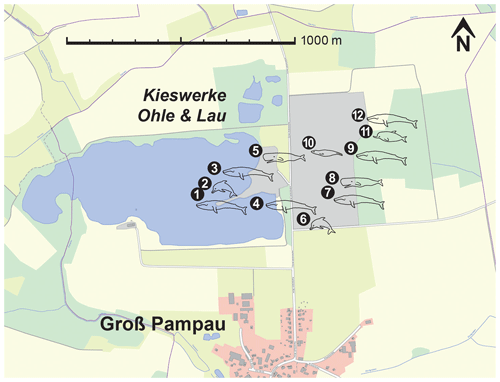
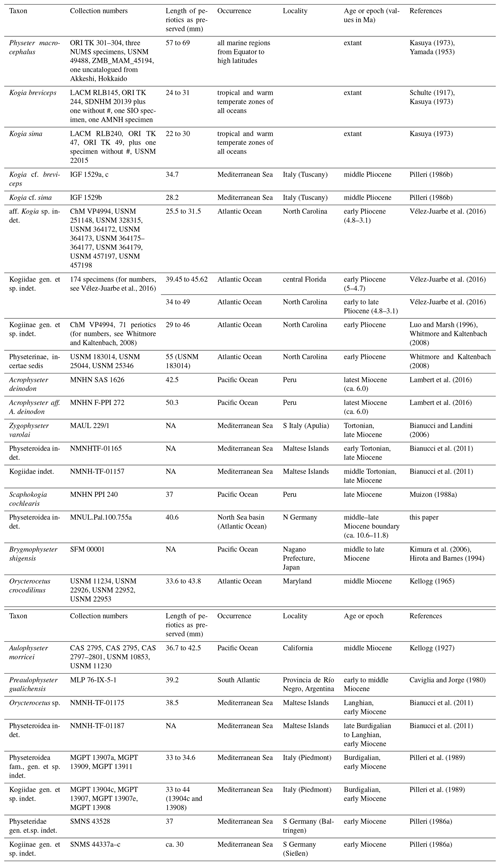
Figure 1Map of the Kieswerke Ohle & Lau company gravel pit. The dots represent significant marine mammal findings that are already published, currently being studied or were mentioned in local media after discovery. (1) Mysticete, found 1984, partial skeleton. (2) Kentriodon hoepfneri Kazár and Hampe, 2014, found in two parts in 1984 and 1989, partial skeleton with nearly complete vertebral column. (3) Praemegaptera pampauensis Behrmann, 1995, found 1989, nearly complete specimen. (4) Praemegaptera pampauensis, found 1993, partial skeleton. (5) Hoplocetus ritzi Hampe, 2006; found 1997; teeth, vertebrae, and bone fragments from a single dislocated skeleton. (6) Kentriodon hoepfneri, found 2000, partial skeleton. (7) Mysticete, found 2012, partial skeleton. (8) Physeteroidea indet., this paper, found 2014, right periotic accompanied by diverse isolated bone fragments. (9) Mysticete, found 2016, partial skeleton. (10) Otariid pinniped; found 2016; ± complete hind limb, bone fragments, teeth. (11) Ziphiidae indet., found 2017, skull. (12) Mysticete, found 2019, partial skeleton. Extract of the “DigitalerAtlasNord” (ETRS89/UTM zone 32N), State Office for Surveying and Geoinformation of Schleswig-Holstein (LVermGeo SH).
For at least 3.5 decades, skeletal elements and teeth of marine mammals have been excavated from the Groß Pampau pit. The findings have been related to Mysticeti, Delphinoidea, and Pinnipedia (Höpfner, 1991; Spaeth and Lehmann, 1992a, b; Moths and Höpfner, 1993; Behrmann, 1995; Lierl, 1995; Hampe, 1999; Moths, 2003; Kazár and Hampe, 2014). Additionally, Hampe (2006) described Hoplocetus ritzi, a new hoplocetine physeterid from Groß Pampau. This and specimens that are no longer verifiable (whereabouts unknown) of Scaldicetus (Abel, 1905; Gripp, 1964) are so far the only fossil remains of physeteroids found in northern Germany. The right periotic of Groß Pampau either might belong to H. ritzi or represents a so-far undescribed species of Physeteroidea found in this pit (Fig. 1).
The extant diversity of physeteroids comprises three species: Physeter macrocephalus Linnaeus, 1758; Kogia breviceps Blainville, 1838; and Kogia sima Owen, 1866. A higher number of extinct taxa extend through the Neogene and even occur already in the late Oligocene of Azerbaijan, from where the oldest physeteroid and presumably one of the oldest cetaceans, Ferecetotherium kelloggi Mchedlidze, 1970, is known. The large fossil record with significant cranial material indicates that Physeteroidea widely diversified during the Miocene (Fordyce and Muizon, 2001; Bianucci and Landini, 2002). Nonetheless, the also-abundant fragmentary or isolated material containing less diagnostic information suggests that the diversity might be even greater than so far considered (e.g. Pilleri, 1986a; Pilleri et al., 1989; Luo and Marsh, 1996; Peters and Monteiro, 2005; Whitmore and Kaltenbach, 2008; Bianucci et al., 2011; Vélez-Juarbe et al., 2016).
The anatomy of extant and extinct odontocetes as currently understood is defined by several combined characters of the skull, mandibles, and ear bones (Fordyce, 1994; Bianucci and Landini, 2006). The tympanic and periotic bones of the odontocete ear form a highly modified complex that is specialized in underwater acoustics and echolocation (e.g. Ketten, 1992, 2000; Luo and Eastman, 1995; Luo and Marsh, 1996; Nummela et al., 2007; Gutstein et al., 2014), with relatively large morphological variability between taxa. This variability in morphological characters and dimensions is often used in the identification of the taxonomic family, genus, and even species of Odontoceti, with preserved ear bones (Yamada, 1953; Kasuya, 1973).
Here, we analyse morphological characteristics that attribute the Groß Pampau periotic to the Physeteroidea yet notably distinguish it from all hitherto-described physeteroid periotic bones, thus suggesting the existence of an undescribed species with a so-far uncertain position in the physeteroid phylogeny.
The bone reported in this work was found by Andreas Malchow, a member of the successful “Grabungsteam”, a group of skilled erratic boulders collectors. These highly motivated self-educated palaeoenthusiasts cooperate closely with the Museum für Natur und Umwelt in Lübeck and the Museum für Naturkunde Berlin and are responsible for most of the discoveries in the Groß Pampau gravel pit. After preparation the bone was measured with an electronic digital Kraftixx caliper, kwb Germany GmbH, Stuhr, and digitally photographed with a Nikon D810, Nikon Corporation, Tokyo, Japan.
The qualitative comparisons of physeteroid periotics undertaken in this work are based on relevant morphological characters obtained from their descriptions and existing photographs, images, or drawn illustrations. As a benchmark, the tympanoperiotic complex of Physeter macrocephalus ZMB_MAM_45194 from the collection of the Museum für Naturkunde Berlin was used for morphological evaluation.
The terminology used here to describe the periotic bone follows Mead and Fordyce (2009) unless otherwise stated.
Among extant and extinct physeteroids relevant and suitable for comparison with the Groß Pampau periotic, the following taxa with sufficiently well-preserved periotic bones have been identified (see collection numbers in Table 2): the macroraptorial physeteroids Acrophyseter deinodon, Acrophyseter aff. A. deinodon, and Acrophyseter robustus (Lambert et al., 2008, 2016); Brygmophyseter shigensis (Hirota and Barnes, 1994; Kimura et al., 2006); and Zygophyseter varolai (Bianucci and Landini, 2006). There are also known several specimens of Physeteroidea indet. from the late Burdigalian of Piedmont and the Burdigalian or Langhian and early Tortonian of the Maltese Islands (Pilleri et al., 1989; Bianucci et al., 2011).
Among Physeteridae, periotics have been described for Aulophyseter morricei (Kellogg, 1927), Orycterocetus crocodilinus Cope, 1867 (Kellogg, 1965), Orycterocetus sp. (Bianucci et al., 2011), Preaulophyseter gualichensis (Caviglia and Jorge, 1980), Physeter macrocephalus (Yamada, 1953; Kasuya, 1973), and a number of Physeterinae gen. et spec. indet. specimens from the Miocene of Antwerp and from the early Pliocene of North Carolina (Peters and Monteiro, 2005; Whitmore and Kaltenbach, 2008), as well as a Physeteridae gen. et spec. indet. specimens from the early Miocene of Baltringen, S Germany (Pilleri, 1986a).
Regarding the family of the Kogiidae, records of periotics exist for the extant Kogia breviceps (Schulte, 1917; Kasuya, 1973) and Kogia sima (Nagorsen, 1985; Kasuya, 1973); Kogia cf. breviceps and Kogia cf. sima (Pilleri, 1986b); Kogia sp. indet. (Vélez-Juarbe et al., 2016); Scaphokogia cochlearis (Muizon, 1988a); specimens of Kogiinae gen. et sp. indet. from the early Miocene of Baltringen and early Pliocene of North Carolina (Pilleri, 1986a; Luo and Marsh, 1996; Whitmore and Kaltenbach, 2008); and specimens of Kogiidae gen. et sp. indet. from the late Burdigalian of Piedmont, the middle Tortonian of the Maltese Islands, the early Pliocene of Florida and the early to late Pliocene of North Carolina (Pilleri et al., 1989; Bianucci et al., 2011; Vélez-Juarbe et al., 2016).
The above-mentioned material was used for the purpose of comparison, except for the cases of A. robustus and the Physeterinae gen. et spec. indet. specimen from the Miocene of Antwerp, which are insufficiently described and thus will not be taken into consideration in this work.
Institutional abbreviations
-
AMNH – American Museum of Natural History,
New York, NY, USA -
CAS – California Academy of Sciences,
San Francisco, CA, USA -
ChM – The Charleston Museum, Charleston, SC, USA
-
IGF – Museo di Storia Naturale, Geologia e Paleontologia, Università degli Studi di Firenze, Florence, Italy
-
LACM – Natural History Museum of Los Angeles
County, Los Angeles, CA, USA -
MAUL – Museo dell'Ambiente, Università del Salento,
Lecce, Italy -
MGPT – Sezione di Paleontologia del Museo Regionale di Scienze Naturali, Turin, Italy
-
MLP – Museo de La Plata, La Plata, Argentina
-
NMNH – National Museum of Natural History,
Mdina, Malta -
MNHN – Muséum National d'Histoire Naturelle,
Paris, France -
MNUL – Museum für Natur und Umwelt,
Lübeck, Germany -
NUMS – Faculty of Medical Sciences, Nagasaki
University, Nagasaki, Japan -
ORI – Atmosphere and Ocean Research Institute,
University of Tokyo, Tokyo, Japan -
SIO – Scripps Institution of Oceanography, University of San Diego, La Jolla, CA, USA
-
SFM – Shiga Fossil Museum, Nagano Prefecture,
Matsumoto, Japan -
SMNS – Staatliches Museum für Naturkunde,
Stuttgart, Germany -
USNM – United States National Museum of Natural History, Smithsonian Institution, Washington, DC, USA
-
ZMB – Zoological Collection, Museum für Naturkunde Berlin, Berlin, Germany
-
Class Mammalia Linnaeus, 1758
-
Order Cetacea Brisson, 1762
-
Suborder Odontoceti Flower, 1867
-
Superfamily Physeteroidea Gray, 1821
-
Physeteroidea, indet.
(Figs. 2, 3, 4)
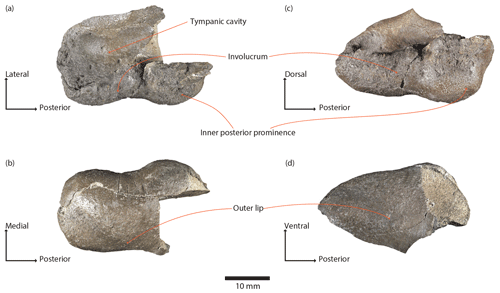
Figure 2Physeteroidea indet., MNUL.Pal.100.755a, fragment of the right tympanic bulla from the upper Langenfeldian (Serravallian–Tortonian boundary) of Groß Pampau. (a) Lateral, (b) medial, (c) dorsal, and (d) ventral view.
Figure 3Physeteroidea indet., MNUL.Pal.100.755a, right periotic from the upper Langenfeldian (Serravallian–Tortonian boundary) of Groß Pampau. (a, b) Dorsal, (c) medial, (d) lateral, (e, f) ventral, (g) anterior, and (h) posterior view.
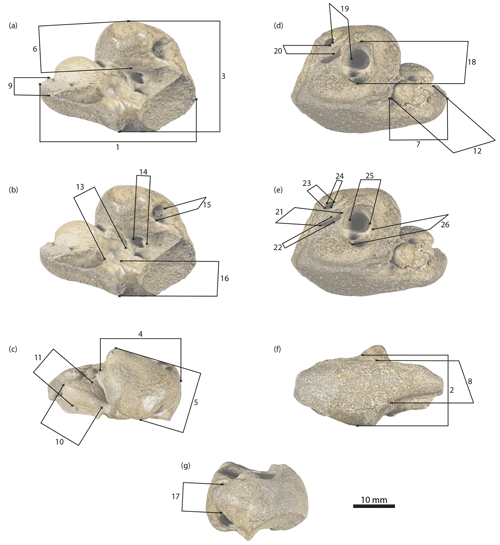
Figure 4Physeteroidea indet., MNUL.Pal.100.755a, measurements of the periotic bone applied in this paper (see Table 1). (a, b) Ventral, (c) medial, (d, e) dorsal, (f) lateral, and (g) posterior view.
Material
One incomplete right periotic (MNUL.Pal.100.755a), one bullar fragment (MNUL.Pal.100.755b; see also comments in the Introduction).
Locality
Groß Pampau, Schleswig-Holstein, northern Germany.
Age
Miocene mica clay deposits underneath Pleistocene gravel; Bolboforma fragori–subfragoris Zone, upper Langenfeldian, about Serravallian–Tortonian boundary, ca. 10.6–11.8 Ma (see Spiegler and Gürs, 1996).
Description
Only the recovered periotic is described in detail here due to the poor preservational status of the other fragmentary bones. The bullar fragment consists of a lower part of the involucrum presenting the spoonlike depressed tympanic cavity. The inner posterior prominence is also determinable – all other parts are broken away (Fig. 2).
The current condition of the periotic bone is incomplete but well preserved (Fig. 3a–h). The bone is composed of a large pars cochlearis and a relatively large body and anterior process. A small posterior portion of the body and the posterior process are lost due to a breakage (indicated by crosshatched area in Fig. 3e, f). The bone tissue is in general compact and dense, although fragile in thin portions.
Measurements of the periotic are shown in Table 1 and Fig. 4a–g.
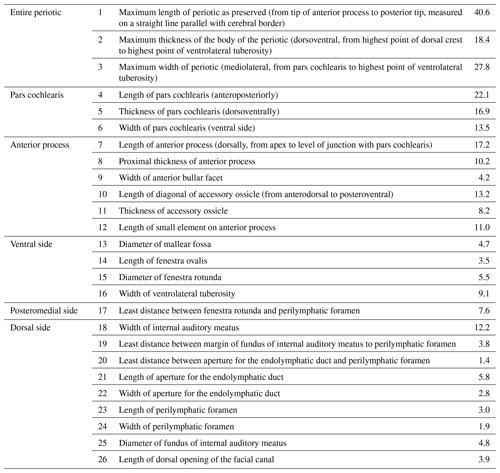
Table 1Measurements (mm) of Physeteroidea indet., MNUL.Pal.100.755a (building upon Kasuya, 1973, and Vélez-Juarbe et al., 2016).
Dorsal view of the periotic (Fig. 3a, b). The large pars cochlearis displays the following shape: the posterior and medial surfaces are flattened; the latter ascends anteriorly; the anterior surface is markedly vaulted. A prominence on the anteromedial edge arises dorsally as the highest point on the dorsal surface.
The internal auditory meatus has a wide ovate shape and is located nearly in the middle of the dorsal surface of the pars cochlearis. It contains, in a funnel-like hollow, the tractus spiralis foraminosus, the foramen singulare, and the area vestibularis inferior. Furthermore, it encloses the small area vestibularis superior and the oval and larger dorsal opening of the facial canal. Both are located in this order laterally to the transverse crest. A septum, the Bill's bar (see definition in Reiss, 2003; Ritsche, 2018), is mediolaterally tilted and separates the area vestibularis superior and the foramen of the facial canal. Both the transverse crest and the Bill's bar are distinctly below the outside rim of the meatus.
While the medial rim is somewhat elevated, the lateral does not protrude noticeably from surrounding bone. The elliptic aperture for the endolymphatic duct lies obliquely on the dorsal surface of the pars cochlearis posterior but very close to the internal auditory meatus. It is separated by a thin bony septum from the smaller and more rounded perilymphatic foramen that lies medially.
Lateral to the aperture for the endolymphatic duct is a rounded protuberance, the dorsal tuberosity, delimited anteriorly by the slightly pronounced posterior rim of the internal auditory meatus.
The suprameatal fossa forms an elongated but shallow depression that is wider posteriorly than anteriorly. It extends laterally to the dorsal tuberosity and internal auditory meatus and medially to the dorsal crest. The latter is a pronounced elevation stretched from a point posterolateral to the dorsal tuberosity towards the anterior. At the level of the area vestibularis superior, the slender crest is oriented more medially and reaches almost the anterior incisure.
An elongated prominence extends laterally from a point posterior to the perilymphatic foramen until it joins the dorsal crest at nearly a right angle forming a small peak. The posterior surface of the periotic body is flattened.
In dorsal view, the rugose tegmen tympani is convex and merges anteriorly into the anterior process. The anterior process is elongated, markedly longer than wide, thin, and shows a slightly pronounced keel as the anterior continuation of the dorsal crest. The lateral part of the anterior process forms a diagonal that rises anteriorly and bends in the ventral direction towards the apex, making the anterior portion of the process rounded. The surface of the anterior process is wrinkled. Prominent dorsal and anterodorsal spines (Muizon, 1988a) are absent, as well as an anteroexternal sulcus.
The pars cochlearis and anterior process meet at nearly a right angle. Two bony elements coalesce to the medial surface of the anterior process. The larger element, the accessory ossicle, is placed more ventrally and has a vaulted shape and an anteriorly pointed edge. The second is a smaller element placed dorsally with an oval but less uniform shape and small protuberances. It covers a large part of the medial surface of the anterior process and a suture between both is visible. Posterior to and at the level of this small element the surface shows a series of grooves that extend ventrally.
Lateral view of the periotic (Fig. 3d). The lateral surface of the tegmen tympani and of the anterior process is slightly rugose and of convex shape. The dorsal margin is delimited by the dorsal crest and its anterior continuation.
The dorsal tuberosity and the pars cochlearis protrude dorsally, the latter with an anteriorly tilted pyramidal shape exposing partially the internal auditory meatus.
The anterior process is thick at the base and tapers anteriorly. Spines on the anterior process are not pronounced; only a very slight protuberance is present and marks the point from where the process narrows out more rapidly in the anterior direction. The ventral outline of the anterior process is a marked concave curvature that accentuates the ventrally pointed apex. Due to the ventral bend of the apex, the anterior margin of the process is rather truncated. The breakage of the outer lip of the tympanic bulla and its suture to the periotic are ventrally visible.
The ventrolateral tuberosity (Luo and Gingerich, 1999) also protrudes ventrally, posterior to the base of the anterior process. From this view, it has a subtriangular outline and an erratically rugose surface. Due to the thinness and curvature of the anterior process, its apex and the ventrolateral tuberosity do not lie on the same horizontal level.
Posterior to the ventrolateral tuberosity, a diagonal edge marks the breakage of the posterior process and a part of the caudal tympanic process remains exposed.
The posterior margin of the periotic body is slightly concave.
Ventral view of the periotic (Fig. 3e, f). The pars cochlearis is slightly anteriorly inclined, and its medial surface is flattened, demonstrating a rectangular outline. The ventral surface is the most convex part of the pars cochlearis, of which the midpoint is flattened at the same time.
The fenestra rotunda is large and almost circular in frontal view. A fissure extends dorsally from its dorsomedial margin towards the most lateral point of the perilymphatic foramen.
Lateral to the fenestra rotunda is the origin of the slightly pronounced, keel-like caudal tympanic process that extends posteriorly and sharpens at the point of breakage.
The fenestra ovalis and laterally the ventral opening of the facial canal are situated posteriorly to the level of the midpoint of the pars cochlearis. The long axis of the fenestra ovalis is oriented anteromedially to posterolaterally. The ventral opening of the facial canal is nearly circular and flows into a shallow depression that extends posteromedially towards the also shallow fossa for the stapedius muscle. This is situated posteriorly to the fenestra ovalis and is flanked medially by the caudal tympanic process. The fossa for the stapedius muscle is also affected by the breakage.
On a keel lateral to the ventral opening of the facial canal rises the circular profile of the incudal process (Luo and Marsh, 1996). Anterior to the incudal process is the mallear fossa, a large circular, shallow depression. The pyramidal ventrolateral tuberosity is elevated laterally between the mallear fossa and incudal process. In this view, its surface and the surface of the periotic body are smooth. A hiatus epitympanicus is not visible due to the breakage posterior to the ventrolateral tuberosity.
The anterior process is thin and tapers towards its apex that forms the anteroventral spine, which is partially damaged. A groove with a medially elevated rim, possibly for the tensor tympani muscle, extends dorsoventrally between the pars cochlearis and accessory ossicle along the anterior incisure towards the ventral opening of the facial canal. The accessory ossicle is vaulted and lies medially to the broken lip of the tympanic bulla. Both cover the anterior process almost entirely, except for a small anterior part that remains uncovered likely due to the breakage of the tympanic bulla.
The lateral outline of the anterior process rises anteriorly, is in the beginning concave, and then becomes slightly convex. The anteroventral spine reaches about the same horizontal level as the ventral opening of the facial canal.
Medial view of the periotic (Fig. 3c). The pars cochlearis has a somewhat triangular outline ventrally rounded. Its ventral protrusion obscures a portion of the fenestra rotunda, and its dorsal protrusion on the anterior edge has an anteriorly tilted pyramidal shape. The opening of the internal auditory meatus, the aperture for the endolymphatic duct, the perilymphatic foramen, and the dorsal crest are only partially visible in this view.
The posterior margin of the pars cochlearis merges into the body of the periotic bone. This surface forms an obtuse angle with the medial surface of the pars cochlearis.
The length of the anterior process comprises more than one-third of the length of the broken periotic bone. The dorsal outline of the anterior process is convex. The small element coalesced to the medial surface is thin and slightly shorter than the accessory ossicle. These have only a little contact with each other at the anterior incisure. The accessory ossicle displays a trapezoid shape and covers the medial surface of the anterior process almost completely. Lateral to this is the broken area of the fused tympanic bulla.
The anteroventral spine of the anterior process is situated dorsally to the ventral edge of the pars cochlearis and is bent so that the anterior bullar facet is facing ventrally and slightly medially.
The aim of this assessment of morphological characters is to comparatively elucidate the relation of the Groß Pampau periotic and other previously described periotics of Physeteroidea, rather than to carry out phylogenetic or functional analyses. The first are impeded by the unclear taxonomic determination of some specimens included in this work, and the second are impeded due to the lack of essential CT-scan data on the internal morphology of the periotic.
4.1 Periotic length
The total length of the periotic as preserved is the measure most frequently reported and is available only for some specimens (see Table 2).
With regard to the compared macroraptorial physeteroids, the Groß Pampau periotic lacking its posterior process approaches the size of Acrophyseter (Lambert et al., 2016), and considering the Physeteridae, it approaches the higher values reported for medium-sized physeterid periotics.
The periotic of Physeter macrocephalus (the largest physeterid) has been described as massive and one of the largest among all odontocete species (Yamada, 1953; Kasuya, 1973). Both P. macrocephalus and one Physeter-like Physeterinae specimen from North Carolina (USNM 183014) exceed the length of the Groß Pampau periotic; this would most likely remain the case even if the periotic were intact. This is also reflected by the maximal dorsoventral height of USNM 183014 (27 mm) in contrast to that of the periotic from Groß Pampau (18.4 mm; Whitmore and Kaltenbach, 2008).
4.2 Dorsal view of the periotic
4.2.1 Pars cochlearis
While the medial surface of the pars cochlearis of the Groß Pampau periotic is rather flattened, the shape of the pars cochlearis has been generally described for macroraptorial physeteroids as raised medially or even showing an almost spherical shape (e.g. Acrophyseter and Zygophyseter varolai; Bianucci and Landini, 2006; Lambert et al., 2016). This is true for Brygmophyseter shigensis only in ventral view, while dorsally, the medial surface of the periotic of this macroraptorial is less flattened than that of the Gross Pampau specimen (Kimura et al., 2006). Periotics of Orycterocetus from the Maltese Islands display a rather high pars cochlearis and Preaulophyseter gualichensis a more globular shape of the pars cochlearis and body of the periotic (Kellogg, 1965; Caviglia and Jorge, 1980). P. macrocephalus has an almost spherically shaped pars cochlearis and thus differs from the condition of the Groß Pampau periotic.
4.2.2 Anterior process
The anterior processes of the periotics of macroraptorial physeteroids, except for Z. varolai (25.3 mm), tend to be shorter (ranging from 6.7 to 11.2 mm) than that of the Groß Pampau periotic (17.2 mm), and some appear to surround the anterior part of the pars cochlearis (e.g. Acrophyseter, Z. varolai, and the physeterid P. gualichensis). In these cases the angle formed by the pars cochlearis and the anterior process at the anterior incisure is highly acute (e.g. Caviglia and Jorge, 1980; Bianucci and Landini, 2006; Lambert et al., 2016). In Z. varolai, a remainder of the anteroexternal sulcus is present in the lateral surface of the anterior process, which is lacking in the Groß Pampau periotic.
Several of the physeteroid periotics display a voluminous and prominent tubercle on the dorsomedial region of the anterior process, often near to the pars cochlearis, which in some cases (e.g. A. deinodon, Acrophyseter aff. A. deinodon) is somewhat longer than the accessory ossicle. As part of the anterior process, the tubercle has a similar surface and influences the overall shape of this structure. The shape and degree of protrusion of the tubercle varies among the specimens, from rounded to more pointy and from small to majorly robust. It is especially developed in the periotics of Acrophyseter aff. A. deinodon, Z. varolai, and B. shigensis (Bianucci and Landini, 2006; Kimura et al., 2006; Lambert et al., 2016), but it is also present in P. gualichensis (Caviglia and Jorge, 1980). This strongly contrasts with the flattened medial and dorsal surfaces of the anterior process of the Groß Pampau periotic, which lacks such a protuberance.
The dorsal surface of the anterior process is wider and flatter in P. macrocephalus than in the Groß Pampau periotic. However, the anterior processes of both periotics do not feature prominent characteristic dorsal or anterodorsal spines. Dorsally to the accessory ossicle in P. macrocephalus, an elongated structure emerges from the anterior process and fuses with the ossicle. Sutures are present between this element and both the anterior process and the ossicle. Its position resembles that of the small element in the Groß Pampau periotic; however, it is less detached from the surrounding structures.
4.2.3 Internal auditory meatus
The relative position of the internal auditory meatus on the pars cochlearis varies among specimens. In the Groß Pampau periotic, as well as in other specimens such as Z. varolai and A. deinodon, the internal auditory meatus is located approximately in the centre (Bianucci and Landini, 2006; Lambert et al., 2016). In the Physeteroidea from the Maltese Islands (NMNH-TF-01165 and 01187), Acrophyseter aff. A. deinodon, and B. shigensis (Kimura et al., 2006; Bianucci et al., 2011; Lambert et al., 2016) the internal auditory meatus is positioned notably more anteriorly, and in one Physeteroidea from Piedmont (MGTP 13907a; Pilleri et al., 1989) it is shifted to a more medial position.
4.2.4 Foramina
In most macroraptorial physeteroid periotics, Aulophyseter, and Orycterocetus (Kellogg, 1927, 1965; Bianucci et al., 2011), as in the Groß Pampau periotic, the aperture for the endolymphatic duct is located externally to the internal auditory meatus.
The condition of the dorsal opening of the facial canal within the internal auditory meatus has been described for Acrophyseter and Z. varolai, P. gualichensis, and for most of the Physeterinae periotics from North Carolina (USNM 183014, USNM 25044, USNM 25346) and coincides with what is observed in the periotic of Groß Pampau. In B. shigensis the dorsal opening of the facial canal is excluded from the meatus (Caviglia and Jorge, 1980; Bianucci and Landini, 2006; Kimura et al., 2006; Whitmore and Kaltenbach, 2008; Lambert et al., 2016).
On the dorsal pars cochlearis of O. crocodilinus and A. morricei periotics, the most anteriorly located opening is the dorsal opening of the facial canal. In contrast, in Orycterocetus sp. as in the Groß Pampau periotic, this foramen is located more centrally and within the internal auditory meatus (Kellogg, 1927, 1965; Bianucci et al., 2011).
P. macrocephalus shows a more laterally located internal auditory meatus in the pars cochlearis, instead of centred as in the Groß Pampau periotic. This peculiarly lodges the aperture for the endolymphatic duct (which is also the case in USNM 183014 from North Carolina; Yamada, 1953; Whitmore and Kaltenbach, 2008), as well as the dorsal opening of the facial canal. The fissure that extends from the fenestra rotunda in the dorsal direction towards the perilymphatic foramen is much deeper in P. macrocephalus than in the Groß Pampau specimen.
4.2.5 Dorsal crest and tegmen tympani
Among physeteroid periotics the dorsal crest is differentiated to a varying extent: low in Acrophyseter and Z. varolai, more pointed in A. morricei and O. crocodilinus, and longer but more massive in Physeter. In some specimens a massive (e.g. Z. varolai), prominent (A. morricei, some O. crocodilinus), or less massive but evident (NMNHTF-01165 from the Maltese Islands) pyramidal process is located posterolaterally to the aperture for the endolymphatic duct (Kellogg, 1927, 1965; Bianucci and Landini, 2006; Bianucci et al., 2011; Lambert et al., 2016). The pyramidal process is a structure initially identified by Luo and Gingerich (1999) in archaeocetes. In contrast, the Groß Pampau periotic displays a particular character combination of a modest dorsal tuberosity and a large suprameatal fossa laterally delimited by a strongly developed dorsal crest pointed at its posterior end.
Periotics of both Orycterocetus and Aulophyseter share with that of Groß Pampau a convex-shaped tegmen tympani (Kellogg, 1927, 1965; Bianucci et al., 2011). In contrast, P. macrocephalus lacks a convex tegmen tympani but displays a remarkably large pyramidal process adjacent to a massive posterior part of the periotic body. A clear separation between pyramidal and superior processes and a well-defined suprameatal fossa as developed in the Groß Pampau periotic are absent here.
4.3 Lateral view of the periotic
4.3.1 Anterior process
A striking difference between the periotic of Groß Pampau and those of macroraptorial physeteroids is evident in lateral and medial views: in Acrophyseter, Z. varolai, and Physeteroidea from the Maltese Islands (NMNHTF-01165 and 01187) and from Piedmont (MGPT 13909) the apex of the anterior process is almost on the same ventral level as or even exceeds the height of the ventrolateral tuberosity (Pilleri et al., 1989; Bianucci and Landini, 2006; Bianucci et al., 2011; Lambert et al., 2016). In B. shigensis the apex of the anterior process does not seem to reach the ventrolateral tuberosity, but the flexure of the anterior process in the ventral direction is considerably stronger than in the Groß Pampau periotic (Kimura et al., 2006).
The anterior process of the periotic of Groß Pampau is strikingly similar to that of P. macrocephalus in its overall shape and disposition to other structures. In this view, in the P. macrocephalus periotic like in that of Groß Pampau, the process emerges from the wide body of the periotic and stretches anteriorly, the dorsal surface remaining straight. A protuberance extending from the ventrolateral tuberosity towards the apex forms the lateral surface of the anterior process of the P. macrocephalus periotic. This crest widens the ventral surface of the process, which is not a condition in the Groß Pampau periotic.
The pars cochlearis of the periotic of P. macrocephalus is dorsally barely exposed when viewed from the lateral plane, due to the larger proportions of the tegmen tympani.
4.3.2 Tegmen tympani
The periotic of P. macrocephalus has in lateral view a “helmet-like” shape (Yamada, 1953), which refers to the semi-triangular shape of the tegmen tympani formed by the configuration of superior, anterior, and posterior processes. The last is strongly bent posteroventrally and forms a right angle with the superior process. The tegmen tympani of the Groß Pampau periotic (lacking the posterior process) displays in this view a similar shape to that of P. macrocephalus.
4.4 Ventral view of the periotic
4.4.1 Pars cochlearis
Specimens of Acrophyseter and Z. varolai share with the one of Groß Pampau a remarkable inclination of the pars cochlearis towards the anterior process, a feature that is also highly pronounced in P. gualichensis, A. morricei, Orycterocetus, and the Baltringen Physeteridae (SMNS 43528; Kellogg, 1927, 1965; Caviglia and Jorge, 1980; Pilleri, 1986a; Bianucci and Landini, 2006; Bianucci et al., 2011; Lambert et al., 2016). In P. macrocephalus the anterior inclination is much stronger than in the Groß Pampau periotic.
The periotics of Acrophyseter and Aulophyseter morricei are similar to the Groß Pampau periotic in that the outline of the pars cochlearis in ventral view presents a posteromedial angulation (Kellogg, 1927; Lambert et al., 2016). This is less pronounced in Oryctocetus crocodilinus (Kellogg, 1965) and Physeter.
Orycterocetus periotics display distinctive characters which are lacking in that of Groß Pampau, for instance, a depressed area near the middle of the ventral pars cochlearis (Kellogg, 1965; Bianucci et al., 2011).
Differences between the periotic of A. morricei and the Groß Pampau specimen include the presence of a nodosity or crest in the middle of the ventromedial face of the first, its fenestra ovalis presenting a crescentic shape and a nodosity partly closing this opening (Kellogg, 1927).
4.4.2 Anterior process
The shapes of macroraptorial physeteroid anterior processes vary between short and pointed (in Acrophyseter), robust and rectangular (e.g. in the Physeteroidea NMNH-TF-01165 from the Maltese Islands), short and blunt or elongated and pointed, and extremely ventrally inclined surrounding the pars cochlearis (in B. shigensis and all three MGTP periotics from Piedmont; Pilleri et al., 1989; Kimura et al., 2006; Bianucci et al., 2011; Lambert et al., 2016). These are all major differences to the elongated anterior process of the Groß Pampau periotic that is here slim, tapers towards the apex, and forms a nearly 90∘ angle at the anterior incisure.
The anterior process in O. crocodilinus periotics is more robust than in the Groß Pampau periotic and more similar in ventral view to A. morricei. Its shape, length, and the angle of the anterior incisure vary notably between specimens, but those showing higher similarity to the Groß Pampau periotic also have a dorsal groove on the anterior process that is absent in the latter. The ventral face of this process in O. crocodilinus is deeply concave like in the Groß Pampau periotic but less flattened from side to side; in some specimens the medial face is also concave (Kellogg, 1927, 1965). The Orycterocetus specimens of the Maltese Islands display a very short anterior process compared to that of the Groß Pampau periotic (Bianucci et al., 2011).
Orycterocetus specimens have in general a less convex lateral face of the anterior process than the Groß Pampau periotic (this is even rectilinear in Orycterocetus of the Maltese Islands; Kellogg, 1965; Bianucci et al., 2011).
The anterior processes of A. morricei and the Groß Pampau periotic differ as the former's exhibits a more massive and globose shape, which can be identified, among other indicators, by the rounded-off lateral face between the ventrolateral tuberosity and apex. The process has been described as the “handle end of a crutch when viewed from in front” (Kellogg, 1927, p. 19), which most likely refers to the presence of a dorsal spine that together with the apex confers to the anterior process such a shape. This condition is not present in the in the Groß Pampau periotic. The ventral face of the anterior process in A. morricei is deeply concave like in the Groß Pampau periotic but less flattened from side to side.
The anterior process of SMNS 43528 from Baltringen is similarly ventrally bent, yet more robust, short, and rounded in than in that of the Groß Pampau specimen.
P. gualichensis also displays a robust anterior process with a strongly convex lateral face (Caviglia and Jorge, 1980).
The angle of the anterior incisure is slightly more acute in P. macrocephalus, and the process thins out in a more gradual way, ending in a more pointed and medially directed apex than in the Groß Pampau periotic, but the arrangement of the anterior process and pars cochlearis is similar in both. Striking is that almost the entire medial length of the anterior process of the periotic of P. macrocephalus is occupied by the large and voluminous accessory ossicle that is strongly fused with the process. Similar to the Groß Pampau periotic, the ossicle in P. macrocephalus is ventrally inclined and is delineated ventrolaterally by a broad contact area of the tympanoperiotic components. In contrast to the ossicle on the periotic of Groß Pampau, in P. macrocephalus it is in close contact over a large area with the globose pars cochlearis, even covering it ventrally.
4.4.3 Ventrolateral tuberosity
In the A. morricei periotic the ventrolateral tuberosity on the base of the anterior process that is more prominent than in the Groß Pampau periotic protrudes laterally and is well visible in dorsal view (Kellogg, 1927). While in Orycterocetus sp. from the Maltese Islands the ventrolateral tuberosity protrudes ventrally more strongly, in other Orycterocetus periotics the ventrolateral tuberosity is even less prominent than in the one of Groß Pampau (Kellogg, 1965; Bianucci et al., 2011). In both Groß Pampau and O. crocodilinus periotics the ventrolateral tuberosity is not visible in dorsal view due to the massive tegmen tympani. This is in some O. crocodilinus specimens more similar in shape to that of the Groß Pampau periotic than in others.
4.4.4 Mallear fossa
The Groß Pampau and other physeteroid periotics display round mallear fossae of different depths, a condition that differs in SMNS 43528 from Baltringen, which has a triangular mallear fossa.
4.4.5 Articulation for the incus
In physeteroids, the articulation for the incus on the ventral body of the periotic displays two different conditions. A fossa is present in Acrophyseter, A. morricei, and O. crocodilinus, while a process can be found in B. shigensis, P. macrocephalus, and the Groß Pampau periotic (Kellogg, 1927, 1965; Yamada, 1953; Kimura et al., 2006; Lambert et al., 2016).
4.5 Medial view of the periotic
4.5.1 Pars cochlearis
The pars cochlearis in Acrophyseter and Z. varolai is dorsoventrally compressed and thus shorter than the body of the periotic, which differs from the condition in the Groß Pampau periotic (Bianucci and Landini, 2006; Lambert et al., 2016).
In specimens of Orycterocetus the ventral face and in A. morricei and SMNS 43528 the posterior face of the pars cochlearis are flattened as in the Groß Pampau periotic (Kellogg, 1927, 1965; Pilleri, 1986a; Bianucci et al., 2011).
4.5.2 Anterior process
The medial surface of the anterior process of Z. varolai is dorsoventrally much larger than the height of its pars cochlearis, which is a major difference from the elongated anterior process of the Groß Pampau periotic.
Like in the Groß Pampau periotic, A. morricei exhibits an irregular groove with small nodosities that traverses the medial face of the anterior process.
5.1 Relation to Physeteroidea
The tympanoperiotic complex of odontocetes is, like in all cetaceans, an exceptionally dense and compact bone (Fleischer, 1975; Ketten, 2000). Although the odontocete ear bones bear a high degree of morphological variability, all characters of the tympanoperiotic complex are derived from an ancestral type (Kasuya, 1973). One of the most striking and distinctive characters, recognized by Luo (1998) as one of two conspicuous bullar apomorphies of the Odontoceti, is the presence of an anteroposteriorly elongated accessory ossicle. This accessory ossicle derives from the anterior process of the tympanic bulla and inserts into the tympanic cavity, making contact with the large and rectangular fovea epitubaria on the anterior process of the periotic bone (Luo, 1998; Bianucci and Landini, 2006; Mead and Fordyce, 2009). The accessory ossicle is developed in early ontogenetic stages and maintains a spherical shape in the adult stages (Luo, 1998). In all physeterids; kogiids; almost all ziphiids; and some specimens of Schizodelphis, platanistids and delphinids, the accessory ossicle is secondarily attached or even fused in real synostosis to the anterior process of the periotic bone (Yamada, 1953; Luo and Marsh, 1996; Luo, 1998; Muizon, 1988b; Mead and Fordyce, 2009). This anterior contact and a posterior syndesmosis are the connective points between the tympanic bulla and periotic bone in Odontoceti (Yamada, 1953). The prominent accessory ossicle that remains strongly fused to the anterior process of the here-described Groß Pampau periotic, despite the breakage of the tympanic bulla, is robust evidence for its relation to the Odontoceti.
We associate the Groß Pampau periotic with the Physeteroidea due to the presence of the following characters: partial fusion of the large accessory ossicle of the tympanic bulla with the anterior process of the periotic that is moved towards the tympanic cavity and covers almost the entire ventral surface of the anterior process of the periotic, resulting in a large, rectangular fovea epitubaria (visible only if the ossicle is separated; elliptical in most other odontocetes and anteroposteriorly compressed in ziphiids), and a very reduced anterior bullar facet near the apex. Additionally, the cochlear portion in the Groß Pampau periotic, as in all physeteroids, is anteriorly inclined (Kasuya, 1973; Bianucci and Landini, 2006; Bianucci et al., 2011). The character described by Kasuya (1973) as “the most conspicuous” of physeteroid ear bones is the large and laminated posterior process of the tympanic bulla that forms a synostosis with the posterior process of the periotic bone, while in other odontocetes the suture remains less firm. A real synostosis attaching firmly the posterior processes of the tympanic bulla and perotic might have been the cause of damage and loss of the posterior process in the Groß Pampau periotic at the time of breakage and separation from the tympanic bulla. The two characters shared by the Groß Pampau specimen and the periotics of the Physeteroidea are the real synostosis by which the accessory ossicle is fused to the anterior process and the lack of an anteroexternal sulcus on its lateral aspect (Luo and Marsh, 1996).
Inconsistencies in the use of technical terminology throughout the literature and in the documentation of measurements of morphological structures and several other characters of the periotic bone that are regarded as diagnostic or bear this potential prevent comparisons that might reveal taxonomically important information and hinder more precise quantitative analyses of size and shape. Since several periotics are damaged to different degrees, we take into account that their measurements do not reflect the real values. Thus, we only cautiously suggest that the Groß Pampau periotic most likely belonged to a medium-sized physeteroid.
In most descriptions of physeteroid periotics the shape of the anterior process and the reciprocal disposition with the pars cochlearis seem to bear a great diagnostic signal. Their particular condition in the Groß Pampau periotic differs substantially from every other described periotic, except for the living P. macrocephalus and A. morricei. However, the combination of morphological characters as a whole is unique in the Groß Pampau periotic and there is no evidence that conclusively associates this specimen with previously described physeteroids.
Kellogg (1927, 1965) made almost identical descriptions of the periotics of A. morricei and O. crocodilinus and highlighted their resemblance to periotics of living sperm whales. Although the occurrence of a high degree of intraspecific variation in periotic morphology is likely (“in some cases very notable”; see Caviglia and Jorge, 1980, p. 366), Luo and Marsh (1996) indicate that Kellogg (1965) misidentified at least two periotics as belonging to Orycterocetus, e.g. USNM 22952, which displays high disparity with the rest and has rather monodontid diagnostic characters. This periotic was hence not taken into consideration in our work.
The differences between Orycterocetus and A. morricei most likely go beyond the ones stated by Kellogg (1927, 1965), and interestingly, of both genera, periotics of A. morricei most closely resemble that of Groß Pampau in their overall appearance and in the general arrangement and proportions of single structures, particularly that of the anterior process and pars cochlearis in relation to each other. However, the most remarkable differences between the A. morricei specimen and the Groß Pampau periotic might constitute the differing shape of their anterior processes.
While Kasuya (1973, p. 64) questionably described the position of the dorsal opening of the facial canal in A. morricei and O. crocodilinus as independent from the internal auditory meatus, Kellogg (1927, 1965) presented two character states: in some specimens, the transverse crest that separates this opening and the tractus spiralis foraminosus is lower than the rim of the internal auditory meatus, while in others the crest is as high as the meatus. In both cases it remains unclear whether the dorsal opening of the facial canal is considered to be part of the meatus or not. However, in the first case, the pyriform rim of the meatus suggests that the dorsal opening of the facial canal is embedded in the internal auditory meatus. In the last case, the shape of the internal auditory meatus remains oviform likely due to the exclusion of the dorsal opening of the facial canal. Based on this we suggest that the degree of similarity between periotics of A. morricei and O. crocodilinus to that of Groß Pampau regarding this character varies between specimens.
In general, the periotic of P. macrocephalus has more pronounced and coarse features and a strikingly larger accessory ossicle than that of Groß Pampau. Despite the enormous difference in size, both periotics show a compelling degree of similarity in several relevant characters. According to Kasuya (1973), the anterior process of the periotic of P. macrocephalus displays the primitive rod shape of odontocetes. Based on their similarity, the anterior process of the Groß Pampau periotic might also fall into this shape category.
Regarding the bony element with an unknown function located dorsally to the accessory ossicle in the Groß Pampau, the condition of P. macrocephalus shows the highest degree of similarity among all other physeteroids included in this comparison. The origin and function of the small bony element of irregular shape and surface that is sutured to the medial surface of the anterior process of the Groß Pampau periotic are uncertain, and only P. macrocephalus shows a slightly similar condition. However, this condition in Physeter has not gained sufficient attention throughout the literature.
The type of breakage on the posteroventral part of the Groß Pampau periotic and the shape of the almost-intact posterior and posterodorsal parts of the body suggest the possibility of a similar posterior process to that of P. macrocephalus that projects ventrally, which would confer to the body of the Groß Pampau periotic a comparable helmetlike shape.
5.2 Relation to Hoplocetus ritzi
The remains of the Hoplocetus ritzi (Hampe, 2006) were discovered in the Bolboforma fragori–subfragoris Zone of the upper Langenfeldian (middle–late Miocene) in Groß Pampau and comprise 13 teeth, eight vertebrae, two pieces of ribs, an acromion, and a phalanx from a single dislocated skeleton. No tympanoperiotic complexes are associated with H. ritzi. This specimen is interpreted as belonging to the macroraptorial non-crown physeteroids, albeit a smaller representative, based on its tooth morphology. The locality of discovery, age, and presumed size of the Groß Pampau periotic coincide with those of H. ritzi, which raises the possibility of their affiliation.
5.3 Relation to Kogiidae
It has been proposed that kogiids and physeterids share two characters of periotic morphology (Luo and Marsh, 1996). Firstly, the articulation of the incus with the periotic bone is via a small incudal process that is an area elevated from the surrounding bone. The validity of this first character, however, is questioned by evidence suggesting that some physeterids have a fossa and some macroraptorial physeteroids have a process for the articulation with the incus (e.g. A. morricei and B. shigensis respectively; see above). The second character is the large size of the accessory ossicle. Personal observations and the lack of consistent data on the relative size of the accessory ossicle do not support the theory that the macroraptorial physeteroids have significantly smaller ossicles. These characters are not reliable to draw conclusions regarding the relation of the Groß Pampau periotic to Kogiidae. There is a certain similarity between some kogiids and the Groß Pampau periotic in regard to the proportion of some structures (e.g. anterior and pars cochlearis in kogiids from Florida and North Carolina; see Vélez-Juarbe et al., 2016). Also, the length of the Groß Pampau periotic falls within the ranges reported for other extinct kogiids. However, due to the following morphological differences we consider it unlikely, that the specimen should be referred to the family Kogiidae, and therefore we do not provide more detailed and thorough comparisons of the Groß Pampau periotic with each of the available kogiid periotics.
Morphological characteristics that have been defined as diagnostic for kogiid periotics include an inflated and rounded apex of the short anterior process and the presence of three well-defined spines on it: anterodorsal, anteroventral, and dorsal (Muizon, 1988a; Luo and Marsh, 1996). These occur in different shapes and magnitudes (for example, see Muizon, 1988a, and Vélez-Juarbe et al., 2016), but when seen as corners, they confer to the anterior process its typical outline of an equilateral triangle (Pilleri et al., 1989). The dorsal face of the anterior process of kogiids is convex and bulbous (Luo and Marsh, 1996), and the process is turned against the pars cochlearis. In regard to these conditions, the Groß Pampau periotic strongly differs from kogiid periotics by having a slender anterior process with less lateral curvature and only one well-defined spine or apex located anteroventrally. The orientation of the posterior process, posteriorly along the long axis of the bone, is a diagnostic character for the Kogiidae (Muizon, 1988a; Luo and Marsh, 1996). This contributes to the typical kogiid condition, in which the posterior bullar facet, the anteroventral spine of the anterior process, and the ventrolateral tuberosity all lie on the same ventral level when observed from lateral view. The massive dorsal tuberosity, which is the highest point on the dorsal surface, together with the dorsal spine of the anterior process, which in some kogiids might reach almost the same height as the dorsal tuberosity, confer to the dorsal face of a kogiid periotic's body a remarkably concave shape. This particular arrangement and shape of structures is lacking in the periotic of Groß Pampau and presumably constitutes the greatest difference from kogiid periotics.
The degree of resemblance with P. macrocephalus and shared characters with A. morricei periotics support our association of the periotic of Groß Pampau with Physeteroidea. All previously described periotics share with the Groß Pampau periotic single morphological characters or a combination of these, yet none of them utterly resembles that of Groß Pampau. Its unique combination of characters suggests three plausible scenarios. Firstly, the possible existence of a so-far undescribed physeteroid species, whose position within the phylogeny of Physeteroidea remains at this point unclear. Secondly, the periotic might also be associated with Hoplocetus ritzi, whose fragments were found in the same depositional area of Groß Pampau years before (Hampe, 2006). And thirdly, the specimen might belong to another, already-described taxon, of which the periotic is currently unknown (see Lambert, 2008).
Our observations revealed that in physeteroid periotics, the shape of the anterior process and the reciprocal disposition with the pars cochlearis are characters that largely vary between taxa and thus bear great diagnostic information.
Functional conclusions, for example, about hearing capabilities, cannot be drawn at this stage. Therefore, comprehensive CT-scan data of the cochlea are one of the essential basics (see Ekdale and Racicot, 2015; Ritsche et al., 2018) and are available so far in detail only for the living sperm whale (Davids et al., 2011) and, very recently, for Acrophyseter deinodon (Galatius et al., 2019).
The original fossils are stored in the collection of the Museum für Natur und Umwelt in Lübeck. No further data sets were used for this article.
Both authors conceived the research, wrote the manuscript, and edited the final version of the text. IMR executed the measurements, processed morphological analyses and comparisons, and prepared Figs. 2–4. OH prepared Fig. 1.
The authors declare that they have no conflict of interest.
We are greatly indebted to Andreas Malchow (Hamburg), who discovered the specimen and provided it for our research. We also thank Gerhard Höpfner (Lübeck) and Reiner Ritz (Bad Bramstedt) for information around the circumstances of the discoveries in Groß Pampau. Hwa Ja Götz (Berlin) provided the photographs of the specimen, and Elke Siebert (Berlin) assisted in constructing Fig. 1. Extremely helpful was the additional preparation of the internal auditory meatus of the periotic by Michele Kaiser (Berlin). We are grateful to Marta Fernández (La Plata, Argentina), who provided high-resolution photographs of the Preaulophyseter periotic for comparison, and Detlef Willborn (Berlin) and David Bohaska (Washington, DC) for access to recent and fossil material likewise for comparative purposes. Many thanks go to Indira Ritsche (Berlin) for informative comments and discussions. We would also like to express our cordial thanks to Mette Elstrup Steeman (Gram, Denmark), Gabriel Aguirre-Fernández (Zurich), and the handling editor, Torsten Scheyer (Zurich), for their valuable suggestions and comments in the review process.
This paper was edited by Torsten Scheyer and reviewed by Mette Steeman and Gabriel Aguirre.
Abel, O.: Les odontocètes du Boldérien (Miocène supérieur) d'Anvers, Mém. Mus. Royal d'Hist. Nat. Belgique, 3, 1–155, 1905.
Behrmann, G.: Der Bartenwal aus dem Miozän von Gr.-Pampau (Schleswig-Holstein), Geschiebekunde aktuell, 11, 119–126, 1995.
Bianucci, G. and Landini, W.: Change in diversity, ecological significance and biogeographical relationships of the Mediterranean Miocene toothed whale fauna, Geobios Mém. Spéc., 24, 19–28, 2002.
Bianucci, G. and Landini, W.: Killer sperm whale: a new basal physeteroid (Mammalia, Cetacea) from the late Miocene of Italy, Zool. J. Linn. Soc., 148, 103–131, 2006.
Bianucci, G., Gatt, M., Catanzariti, R., Sorbi, S., Bonavia, C. G., Curmi, R., and Varola, A.: Systematics, biostratigraphy and evolutionary pattern of the Oligo-Miocene marine mammals from the Maltese Islands, Geobios, 44, 549–585, 2011.
Blainville, H. M. D. de: Sur les cachalots, Ann. françaises étrangères Anat. Physiol. appl. Médicine Hist. natur., 2, 335–337, 1838.
Brisson, A. D.: Regnum animale in Classes IX. distributum, sive Synopsis methodica sistens generalem Animalium distributionem in Classes IX, & duarum primarum Classicum, Quadrupedum scilicet & Cetaceorum, particularem divisionem in Ordines, Sectiones, Genera & Species, Haak, Leiden, 296 pp., 1762.
Caviglia, S. E. and Jorge, R. E.: Preaulophyseter gualichensis gen. et sp. nov. (Cetacea; Physeteridae), en el Terciario marino de Rio Negro, Republica Argentina, Actas del Segundo Congreso Argentino de Paleontología y Bioestratigrafía y Primer Congreso Latinoamericano de Paleontología, Buenos Aires 1978, 363–370, 1980.
Cope, E. D.: An addition to the vertebrate fauna of the Miocene period, with a synopsis of the extinct Cetacea of the United States, P. Acad. Nat. Sci. Phila., 19, 138–156, 1867.
Davids, T., Mount, R. J., Blaser, S., Campisi, P., and Harrison, R. V.: CT study of acoustic signal pathway through the middle ear of the sperm whale (Physeter macrocephalus), Canadian Acoustics, 39, 13–17, 2011.
Ekdale, E. G. and Racicot, R. A.: Anatomical evidence for low frequency sensitivity in an archaeocete whale: comparison of the inner ear of Zygorhiza kochii with that of crown Mysticeti, J. Anat., 226, 22–39, 2015.
Fleischer, G.: Über das spezialisierte Gehörorgan von Kogia breviceps (Odontoceti), Z. Säugetierkd., 40, 89–102, 1975.
Flower, W. H.: Description of the skeleton of Inia geoffrensis and of the skull of Pontoporia blainvillii, with remarks on the systematic position of these animals in the order Cetacea, Trans. Zool. Soc. London, 6, 87–116, 1867.
Fordyce, R. E.: Waipatia maerewhenua, new genus and new species (Waipatiidae, new family), an archaic late Oligocene dolphin (Cetacea: Odontoceti: Platanistoidea) from New Zealand, in: Contributions in Marine Mammal Paleontology Honoring Frank C. Whitmore Jr., edited by: Berta, A. and Deméré, T. A., Proc. San Diego Soc. Nat. Hist., 29, 147–176, 1994.
Fordyce, R. E. and Muizon, C. de: Evolutionary history of cetaceans: a review, in: Secondary Adaptation of Tetrapods to Life in Water, edited by: Mazin, J.-M. and Buffrénil, V. de, Dr. Friedrich Pfeil, Munich, 169–233, 2001.
Galatius, A., Olsen, M. T., Steeman, M. E., Racicot, R. A., Bradshaw, C. D., Kyhn, L. A., and Miller, L. A.: Raising your voice: evolution of narrow-band high-frequency signals in toothed whales (Odontoceti), Biol. J. Linn. Soc., 126, 213–224, 2019.
Gray, J. E.: On the natural arrangement of vertebrose animals, The London Medical Repository, 15, 296–310, 1821.
Gripp, K.: Erdgeschichte von Schleswig-Holstein, Wachtholz, Neumünster, 411 pp., 1964.
Gutstein, C. S., Figueroa-Bravo, C. P., Pyenson, N. D., Yury-Yañez, R. E., Cozzuol, M. A., and Canals, M.: High frequency echolocation, ear morphology, and the marine–freshwater transition: A comparative study of extant and extinct toothed whales, Palaeogeogr. Palaeocl., 400, 62–74, 2014.
Hampe, O.: Bestandsaufnahme der Walfauna (Mammalia: Cetacea) aus dem untersten Obermiozän (oberes Langenfeldium) von Groß Pampau (Schleswig-Holstein), Ber. Ver. “Natur und Heimat” Naturhist. Mus. Lübeck, 25/26, 87–107, 1999.
Hampe, O.: Middle/late Miocene hoplocetine sperm whale remains (Odontoceti: Physeteridae) of North Germany with an emended classification of the Hoplocetinae, Foss. Rec., 9, 61–86, https://doi.org/10.1002/mmng.200600002, 2006.
Hirota, K. and Barnes, L.G.: A new species of middle Miocene sperm whale of the genus Scaldicetus (Cetacea; Physeteridae) from Shiga-mura, Japan, Isl. Arc, 3, 453–472, 1994.
Höpfner, G.: Wale und Haie – aus der Urzeit aufgetaucht, Z. Nat. Landeskd. Schleswig-Holstein und Hamburg, 98, 245–253, 1991.
Kasuya, T.: Systematic consideration of recent toothed whales based on the morphology of tympano-periotic bone, Sci. Rep. Whales Res. Inst., 25, 1–103, 1973.
Kazár, E. and Hampe, O.: A new species of Kentriodon (Mammalia, Odontoceti, Delphinoidea) from the middle/late Miocene of Groß Pampau (Schleswig-Holstein, North Germany), J. Vert. Paleont., 34, 1216–1230, 2014.
Kellogg, R.: Study of the skull of a fossil sperm-whale from the Temblor Miocene of Southern California, in: Additions to the palaeontology of the Pacific coast and Great Basin regions of North America, edited by: Kellogg, R., Merriam, J. C., Stock, C., Chaney, R. W., and Mason, H. L., Contr. Palaeont. Carnegie Inst. Washington, 346, 3–24, 1927.
Kellogg, R.: The Miocene Calvert sperm whale Orycterocetus, in: Fossil marine mammals from the Miocene Calvert Formation of Maryland and Virginia, Part 2, Bull. U.S. Nat. Mus., 247, 47–63, 1965.
Ketten, D. R.: The cetacean ear: form, frequency, and evolution, in: Marine Mammal Sensory Systems, edited by: Thomas, J. A., Kastelein, R. A., and Supin, A. Y., Springer, Boston, 53–75, 1992.
Ketten, D. R.: Cetacean Ears, in: Hearing by Whales and Dolphins, edited by: Au, W., Fay, R., and Popper, A., Springer, New York, 43–108, 2000.
Kimura, T., Hasegawa, Y., and Barnes, L. G.: Fossil sperm whales (Cetacea, Physeteridae) from Gunma and Ibaraki prefectures Japan; with observations on the Miocene fossil sperm whale Scaldicetus shigensis Hirota and Barnes, 1995, Bull. Gunma Mus. Nat. Hist., 10, 1–23, 2006.
Lambert, O.: Sperm whales from the Miocene of the North Sea: a re-appraisal, Bull. Inst. royal Sci. nat. Belgique, Sci. Terre, 78, 277–316, 2008.
Lambert, O., Bianucci, G., and Muizon, C. de: A new stem-sperm whale (Cetacea, Odontoceti, Physeteroidea) from the latest Miocene of Peru, C. R. Palevol, 7, 361–369, 2008.
Lambert, O., Bianucci, G., and Muizon, C. de: Macroraptorial sperm whales (Cetacea, Odontoceti, Physeteroidea) from the Miocene of Peru, Zool. J. Linn. Soc., 179, 404–474, 2016.
Lierl, H. J.: Ein miozäner Robbenrest (Hüftbein von Phoca sp.) aus dem Geschiebe von Groß Pampau (Kreis Herzogtum Lauenburg), Geschiebekunde aktuell, 11, 3–10, 1995.
Linnaeus, C.: Systema naturae per Regna tria naturae, secundum Classes, Ordines, Genera, Species, cum Characteribus, Differentiis, Synonymis, Locis. Tomus I. Editio Decima, Reformata, Laurentii Salvii, Stockholm, 824 pp., 1758.
Luo, Z.: Homology and transformation of cetacean ectotympanic structures, in: The Emergence of Whales, edited by: Thewissen, J. G. M., Plenum Press, New York, 269–301, 1998.
Luo, Z. and Eastman, E. R.: Petrosal and inner ear of a squalodontoid whale: implications for evolution of hearing in odontocetes, J. Vertebr. Paleontol., 15, 431–442, 1995.
Luo, Z. and Gingerich, P. D.: Terrestrial Mesonychia to aquatic Cetacea: transformation of the basicranium and evolution of hearing in whales, Univ. Michigan. Pap. Paleont., 31, 1–98, 1999.
Luo, Z. and Marsh, K.: Petrosal (periotic) and inner ear of a Pliocene kogiine whale (Kogiinae, Odontoceti): implications on relationships and hearing evolution of toothed whales, J. Vertebr. Paleontol., 16, 328–348, 1996.
Mchedlidze, G. A.: Nekotorye obshchie cherti istorii kitoobraznykh. Chast' 1, Akademia Nauk Gruzinskoi S.S.R., Institut Paleobiologii, Tbilisi, Georgia, Metsniereba, 1970.
Mead, J. G. and Fordyce, R. E.: The therian skull: a lexicon with emphasis on the Odontocetes, Smithsonian Contr. Zool., 1–249, 2009.
Moths, H.: Fossile Meeresschildkrötenreste und Hüft- und Schienbein einer Robbe aus dem Tertiär Norddeutschlands, Geschiebesammler, 36, 83–99, 2003.
Moths, H. and Höpfner, G.: Ältester Nachweis des Rauhzahndelphines Steno sp. im Glimmerton des Langenfeldiums (Obermiozän) von Groß Pampau, Geschiebesammler, 26, 151–160, 1993.
Muizon, C. de: Les vertébrés fossiles de la formation Pisco (Pérou). Troisième partie: Les odontocètes (Cetacea, Mammalia) du Miocène, Inst. Français Études Andines Mém., 78, 1–244, 1988a.
Muizon, C. de: Le polyphylétisme des Acrodelphidae, odontocètes longirostres du Miocène européen, Bull. Mus. Nat. Hist. Natur., Sect. C, Sci. terre, paléontol., géol., minéral., 10, 31–88, 1988b.
Nargosen, D.: Kogia simus, Mammalian Species, 239, 1–6, 1985.
Nummela, S., Thewissen, J. G. M., Bajpai, S., Hussain, T., and Kumar, K.: Sound transmission in archaic and modern whales: Anatomical adaptations for underwater hearing, Anat. Rec., 290, 716–733, 2007.
Owen, R.: On some Indian Cetacea collected by Sir Walter Elliot, Trans. Zool. Soc. London, 6, 171–174, 1866.
Peters, N. and Monteiro, H.: A small sperm whale (Cetacea: Odontoceti, Physeteridae) from the Miocene of Antwerp, Deinsea, 11, 87–102, 2005.
Pilleri, G.: The Cetacea of the Western Paratethys (Upper Marine Molasse of Baltringen), Hirnanat. Inst., Berne, 70 pp., 1986a.
Pilleri, G.: Pygmy sperm whales (Kogia) in the Italian Pliocene, Invest. Cetacea, 18, 133–153, 1986b.
Pilleri, G., Gihr, M., and Kraus, C.: Odontoceti (Mammalia: Cetacea) from the lower Miocene of Rosignano, Piedmont, North Italy, Invest. Cetacea, 22, 198–291, 1989.
Reiss, G.: Gehör- und Gleichgewichtsorgan, Organum vestibulocochleare, in: Waldeyer – Anatomie des Menschen, 17th Edn., edited by: Fanghänel, J., Pera, F., Anderhuber, F., and Nitsch, R., de Gruyter, Berlin, 593–628, 2003.
Ritsche, I.: The relationship of cochlear morphology and hearing frequencies in cetaceans with emphasis on the occurrence of infrasonic sensitivity in baleen whales, and the classification of mysticete periotic bones from the Miocene of Bocholt, PhD thesis, Humboldt-Universität zu Berlin, Germany, 153 pp., 2018.
Ritsche, I. S., Fahlke, J. M., Wieder, F., Hilger, A., Manke, I., and Hampe, O.: Relationships of cochlear coiling shape and hearing frequencies in cetaceans, and the occurrence of infrasonic hearing in Miocene Mysticeti, Foss. Rec., 21, 33–45, https://doi.org/10.5194/fr-21-33-2018, 2018.
Schulte, H. V. W.: The skull of Kogia breviceps Blainv, B. Am. Mus. Nat. Hist., 37, 361–404, 1917.
Spaeth, C. and Lehmann, U.: Über Bartenwale im nordwesteuropäischen Tertiär, Teil 1, Fossilien, 9, 13–23, 1992a.
Spaeth, C. and Lehmann, U.: Über Bartenwale im nordwesteuropäischen Tertiär, Teil 2, Fossilien, 9, 81–91, 1992b.
Spiegler, D. and Gürs, K.: Der miozäne Glimmerton von Groß Pampau, Schleswig-Holstein (Mollusken, Foraminiferen und Bolboformen), Meyniana, 48, 135–164, 1996.
Vélez-Juarbe, J., Wood, A. R., and Pimiento, C.: Pygmy sperm whales (Odontoceti, Kogiidae) from the Pliocene of Florida and North Carolina, J. Vertebr. Paleontol., 36, e1135806, https://doi.org/10.1080/02724634.2016.1135806, 2016.
Whitmore Jr., F.C. and Kaltenbach, J. A.: Neogene Cetacea of the Lee Creek Phosphate Mine, North Carolina, in: Geology and Paleontology of the Lee Creek Mine, North Carolina, IV, edited by: Ray, C. E., Bohaska, D. J., Koretsky, I. A., Ward, L. W., and Barnes, L. G., Virginia Mus. Nat. Hist. Spec. Publ., 14, 181–269, 2008.
Yamada, M.: Contibution to the anatomy of the organ of hearing of whales, Sci. Rep. Whales Res. Inst., 8, 1–79, 1953.